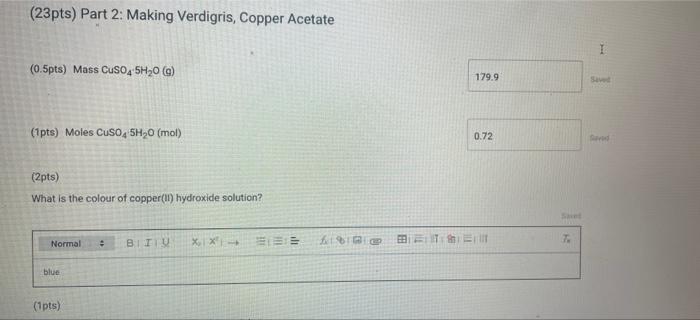
(23pts) Part 2: Making Verdigris, Copper Acetate (0.5pts) Mass CuSO4 5H0 (g) (1pts) Moles CuSO4 5H0...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
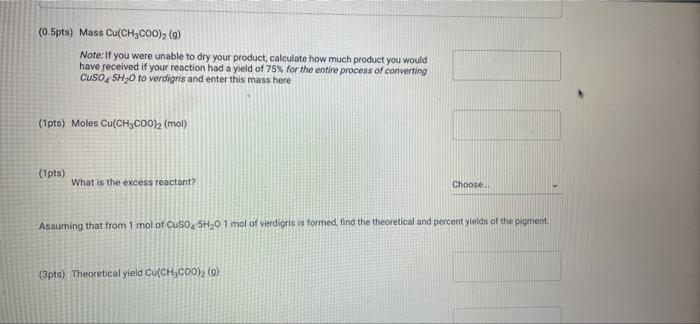
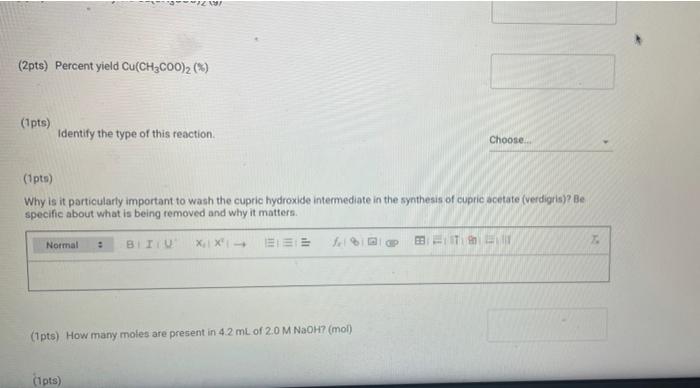
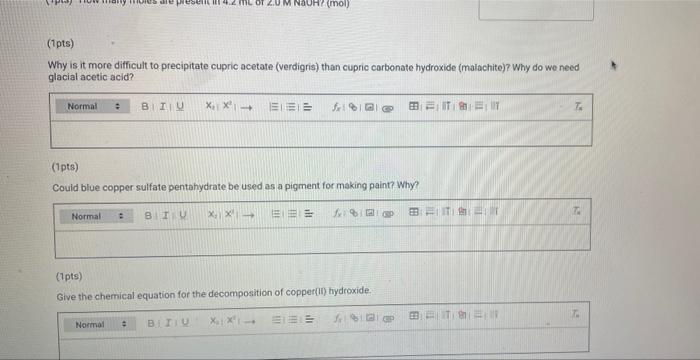
(23pts) Part 2: Making Verdigris, Copper Acetate (0.5pts) Mass CuSO4 5H₂0 (g) (1pts) Moles CuSO4 5H₂0 (mol) (2pts) What is the colour of copper(11) hydroxide solution? Normal blue (1pts) # BITU E 6853 TE 179.9 0.72 Savet T I Saved Seved (0.5pts) Mass Cu(CH₂COO)2 (9) Note: If you were unable to dry your product, calculate how much product you would have received if your reaction had a yield of 75% for the entire process of converting CuSO4 SH₂O to verdigris and enter this mass here (1pts) Moles Cu(CH₂C00)2 (mol) (1pts) What is the excess reactant? 60 (3pts) Theoretical yield Cu(CH₂CO0)₂2 (0) Choose... Assuming that from 1 mol of CuSO, SH₂0 1 mol of verdigris is formed, find the theoretical and percent yields of the pigment. (2pts) Percent yield Cu(CH3COO)2 (%) (1pts) MINI Identify the type of this reaction. Normal : (1pts) Why is it particularly important to wash the cupric hydroxide intermediate in the synthesis of cupric acetate (verdigris)? Be specific about what is being removed and why it matters. BIU X₁ X² → (1pts) How many moles are present in 4.2 mL of 2.0 M NaOH? (mol) (1pts) Choose.... BET es are preselil 4.2 mL of 2.0 M NaOH? (mol) (1pts) Why is it more difficult to precipitate cupric acetate (verdigris) than cupric carbonate hydroxide (malachite)? Why do we need glacial acetic acid? Normal ÷ BIU X₁ X²4 Normal (1pts) Could blue copper sulfate pentahydrate be used as a pigment for making paint? Why? EEE : BIU X, X→ Normal : BIZU EEE 18 (1pts) Give the chemical equation for the decomposition of copper(1) hydroxide. X₁ X² BEITSE BES f BT T Ta 72 (23pts) Part 2: Making Verdigris, Copper Acetate (0.5pts) Mass CuSO4 5H₂0 (g) (1pts) Moles CuSO4 5H₂0 (mol) (2pts) What is the colour of copper(11) hydroxide solution? Normal blue (1pts) # BITU E 6853 TE 179.9 0.72 Savet T I Saved Seved (0.5pts) Mass Cu(CH₂COO)2 (9) Note: If you were unable to dry your product, calculate how much product you would have received if your reaction had a yield of 75% for the entire process of converting CuSO4 SH₂O to verdigris and enter this mass here (1pts) Moles Cu(CH₂C00)2 (mol) (1pts) What is the excess reactant? 60 (3pts) Theoretical yield Cu(CH₂CO0)₂2 (0) Choose... Assuming that from 1 mol of CuSO, SH₂0 1 mol of verdigris is formed, find the theoretical and percent yields of the pigment. (2pts) Percent yield Cu(CH3COO)2 (%) (1pts) MINI Identify the type of this reaction. Normal : (1pts) Why is it particularly important to wash the cupric hydroxide intermediate in the synthesis of cupric acetate (verdigris)? Be specific about what is being removed and why it matters. BIU X₁ X² → (1pts) How many moles are present in 4.2 mL of 2.0 M NaOH? (mol) (1pts) Choose.... BET es are preselil 4.2 mL of 2.0 M NaOH? (mol) (1pts) Why is it more difficult to precipitate cupric acetate (verdigris) than cupric carbonate hydroxide (malachite)? Why do we need glacial acetic acid? Normal ÷ BIU X₁ X²4 Normal (1pts) Could blue copper sulfate pentahydrate be used as a pigment for making paint? Why? EEE : BIU X, X→ Normal : BIZU EEE 18 (1pts) Give the chemical equation for the decomposition of copper(1) hydroxide. X₁ X² BEITSE BES f BT T Ta 72
Expert Answer:
Answer rating: 100% (QA)
1Moles of CuSO45H2O mol 072 mols 2 CopperII hydroxide solution is typically bluegreen We need the ba... View the full answer

Related Book For 

Organic Chemistry
ISBN: 9788120307209
6th Edition
Authors: Robert Thornton Morrison, Robert Neilson Boyd
Posted Date:
Students also viewed these chemistry questions
-
What is the colour of the universal indicator paper? What is the pH? Explain your observations.
-
What would be your reaction if you were told you were not hired based on your responses to this questionnaire?
-
A chemical reaction has a theoretical yield of 19.98 g and a percent yield of 88.40%. What is the actual yield?
-
Three friends are choosing a restaurant for dinner. Here are their preferences: a. If the three friends use a Borda count to make their decision, where do they go to eat? b. On their way to their...
-
Assume that the Rocky Mountain Outfitters customer support system will be developed as described in your answer to question 1. Assume that 14 people are available for programming and testing. What...
-
An electron in a cathode-ray tube is accelerated through a potential difference of 10 kV, then passes through the 2.0 cm-wide region of uniform magnetic field in FIGURE P29.60. What field strength...
-
Apex Electrical Supply, Inc., purchased inventory for \($2,000\) and also paid \($125\) freight to have the inventory delivered. Apex Electrical Supply, Inc., returned \($500\) of the goods to the...
-
Total wages and salaries for the month of January were $50,000. Because it is January, no employee has yet reached the FICA tax cap amount, so the full FICA tax percentage is applicable to the entire...
-
a) Marcia's Leather Knits makes and sells leather jackets. Prior to starting 2021, Marcia made flexible budgets for 900, 1,200, 1,500 sales volume levels. Given the following budget and actual data,...
-
In cell C11, construct a formula to calculate the annual projected street maintenance costs for the year 2025 after the projected increase of 5% is applied. Use absolute cell references as necessary....
-
How managers supporting their employees can improve the efficiency of an organization. And what are some strategies a manager can use to show support and have effective relations?
-
Gross profit, operating profit, and net profit; the three most important profitability ratios. Business owners and managers may find varied usefulness of the other ratios calculated from the income...
-
5) It costs a company c(x) = -0.004x2 + 500x + 50000 to make its product. Find the marginal cost and use it to find how fast the cost is increasing when x = 2000 units. (4 points)
-
1. Place 0.5 g of well-powdered 3-nitrobenzoic acid and a stirring bar into a 10 mL round bottom flask. (The powdering is best done with a 25 mL Erlenmeyer and smooth surfaced weighing paper.) 2....
-
Identify and define at least one ethical framework that the company, Equifax, apparently employed in making its decision in that ethical dilemma. This is during the time that Equifax had a data...
-
An ideal gas initially at 325 K undergoes an isobaric expansion at 2.50 kPa. The volume increases from 1.00 m to 3.00 m and 12.0 kJ is transferred to the gas by heat. (a) What is the change in...
-
write a literature review on the synthesis of flexible latent heat storage material and examination of its thermal performances?
-
Question 2 For an n x n matrix A = form) via (aij)
-
In the context of the previous two problems, with no dividends, compute the price of the chooser option, for which the holder can choose at time t1 = 0.25 years whether to hold the call or the put...
-
Complete combustion of a hydrocarbon causes the carbon atoms in the hydrocarbon molecule to be oxidised to their highest oxidation state, +4. See the experiment which shows the complete combustion of...
-
What did you observe when you added the soap to the hard water and soft water ? Explain your observations.
-
Calculate the residual enthalpy and entropy for carbon dioxide at \(393 \mathrm{~K}\) and \(12 \mathrm{MPa}\) using any equation of state.
-
Prove that \(d S=\frac{C_{P}}{T} d T-\beta V d P\).
-
What assumptions are involved in the Clausius-Clapeyron equation?
Coordinate Metrology Accuracy Of Systems And Measurements 1st Edition - ISBN: 3662484633 - Free Book

Study smarter with the SolutionInn App


