25.0 cm of a solution of sodium hydroxide solution required 21.5 cm of 0.100 mol/dm sulfuric...
Fantastic news! We've Found the answer you've been seeking!
Question:
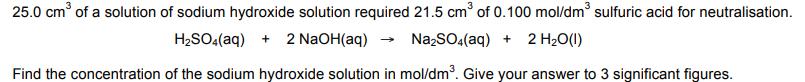
![]()
Transcribed Image Text:
25.0 cm³ of a solution of sodium hydroxide solution required 21.5 cm³ of 0.100 mol/dm³ sulfuric acid for neutralisation. H₂SO4 (aq) + 2 NaOH(aq) → Na₂SO4(aq) + 2 H₂O(1) Find the concentration of the sodium hydroxide solution in mol/dm³. Give your answer to 3 significant figures. Find the concentration of the sodium hydroxide solution in g/dm³. Give your answer to 3 significant figures. 25.0 cm³ of a solution of sodium hydroxide solution required 21.5 cm³ of 0.100 mol/dm³ sulfuric acid for neutralisation. H₂SO4 (aq) + 2 NaOH(aq) → Na₂SO4(aq) + 2 H₂O(1) Find the concentration of the sodium hydroxide solution in mol/dm³. Give your answer to 3 significant figures. Find the concentration of the sodium hydroxide solution in g/dm³. Give your answer to 3 significant figures.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A solution of sodium hydroxide of density 1650 kg/m3 and viscosity 50 mN s/m2 is agitated by a propeller mixer of 0.5 m diameter in a tank of 2.28 m diameter, and the liquid depth is 2.28 m. The...
-
What salt is formed when a solution of sodium hydroxide is neutralized by sulfuric acid? Give the equation of the process.
-
Determine the molar enthalpy of a solution of sodium nitrate, if dissolving 8.95 g of the salt in 104.52 g of water in a simple calorimeter causes the water temperature to drop from 22.6 C to18.2 C
-
Tell whether the given side lengths form a right triangle. 8, 10, and 14
-
Choose one of the major storage sites of the body for toxic substances. Identify a substance that is stored in the tissue and the mechanisms that would affect the transport and absorption of the...
-
In Problems 57, compute the value of the given expression. C(11, 5)
-
Number of bolts in flange coupling should not be less than: (a) 2 (b) 3 (c) 4 (d) 8
-
Journal entries for the issuance of capital stock. Prepare journal entries to record the issuance of capital stock in each of the following independent cases. You may omit explanations for the...
-
If information is reliable, does that guarantee its credibility as well?
-
Lucy loves to play the Hop, Skip, and Jump game. Given an N*M matrix and starting from the cell (1,1), her challenge is to hop in an anti-clockwise direction and skip alternate cells. The goal is to...
-
6. Let A 0 C R which is bounded from below. Prove that inf(A) is the unique real number y satisfying: (a) for all a A, y a, and (b) for each e O there exists a A such that a < y + e.
-
What is the planned monthly EOM for July if the BOM for the next month is the EOM. Example, for February, the EOM inventory would be the BOM for March?
-
.What is meant by the concept of shared and non-shared environmental experiences ?
-
a) List and briefly describe the five conflict managment styles b)State when and how to use five conflict management styles.
-
Can you answer the question related to the preparation of the cash flow statement with explanation of each calculation?
-
Some would argue that fixed costs cannot be controlled by department managers so are not relevant for management decision making, hence variable costing in the only important one. What would be your...
-
Now, assume that Zoe's savings institution modifies the terms of her account and agrees to pay 7% in compound interest on her $900 balance. All other things being equal. How much money will Zoe have...
-
The trade-off theory relies on the threat of financial distress. But why should a public corporation ever have to land in financial distress? According to the theory, the firm should operate at the...
-
A pupil carried out an experiment in a fume cupboard to find out how electricity affected different substances. Some of the substances were in aqueous solution, others were in the molten state....
-
Explain the following. a. Ethene is called an unsaturated hydrocarbon. b. The cracking of larger alkanes into simple alkanes and alkenes is important to the petrochemical industry. c. The conversion...
-
Explain the following. a. Chemical plants that produce sulfuric acid are often located on the coast. b. Even though more sulfuric acid could be produced using high pressures, normal atmospheric...
-
In what ways do you believe providing special work arrangements or accommodations for employees impacts employee motivation? How does it help? How does it hurt?
-
What would be the most effective option to increase employee motivation to stay and reduce the driver turnover rate? Why do you believe this option will be effective?
-
How else might the manager have handled the situation to prevent potential issues, including a negative impact on the teams performance?

Study smarter with the SolutionInn App



