2Na (s) +2H 2 O (l) 2NaOH (aq) +H 2(g) In this chemical equation, H 2 O...
Fantastic news! We've Found the answer you've been seeking!
Question:
2Na(s) +2H2O(l)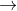 2NaOH(aq) +H2(g)
2NaOH(aq) +H2(g)
In this chemical equation, H2O is the oxidizingagent. I am confused because on the reactant side H2Ohas an oxidation number of 0. How can we tell that it has reducedif there is no H2O to compare to on the product side? Iunderstand that a whole compound can be a reducing or oxidizingagent but how can we say that it is the oxidizing agent if we breakit apart on the product side?
Related Book For 

Posted Date:




