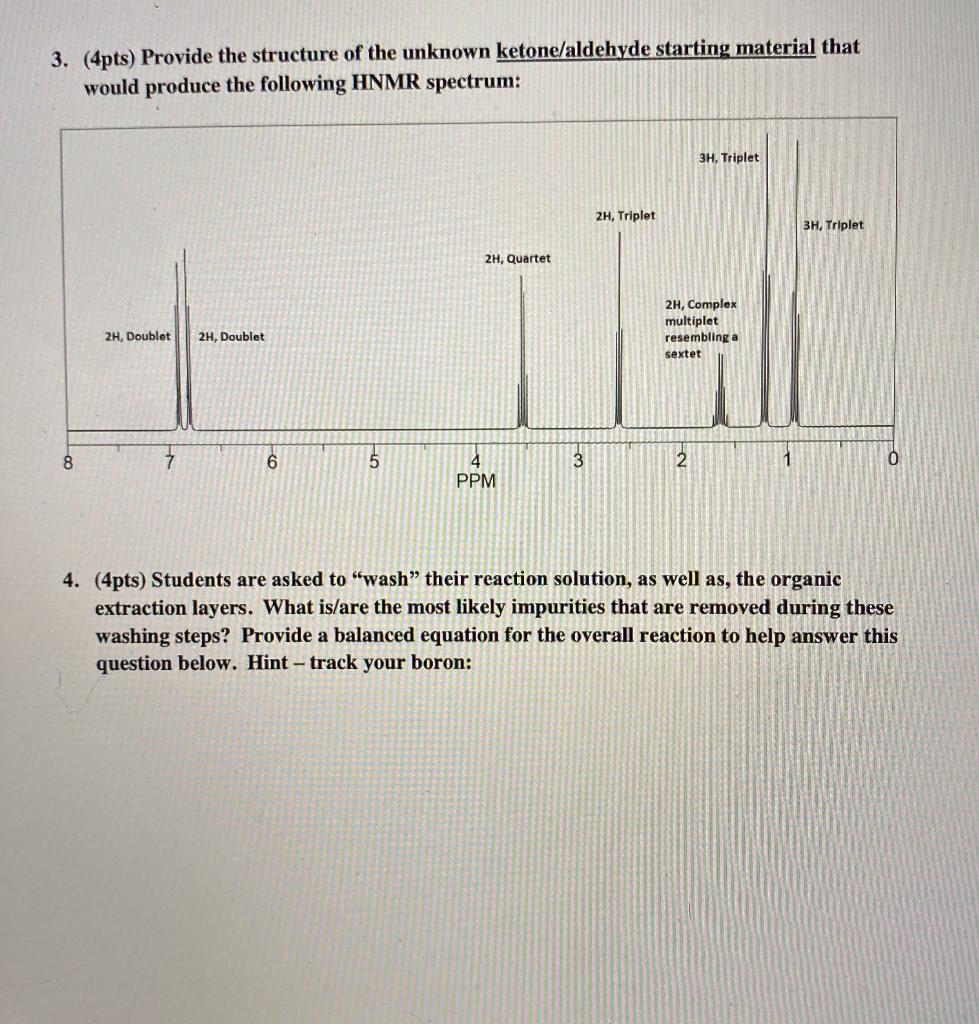
3. (4pts) Provide the structure of the unknown ketone/aldehyde starting material that would produce the following...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
3. (4pts) Provide the structure of the unknown ketone/aldehyde starting material that would produce the following HNMR spectrum: 8 2H, Doublet 2H, Doublet 6 5 2H, Quartet 4 PPM 3 2H, Triplet 3H, Triplet 2H, Complex multiplet resembling a sextet 2 1 3H, Triplet 4. (4pts) Students are asked to "wash" their reaction solution, as well as, the organic extraction layers. What is/are the most likely impurities that are removed during these washing steps? Provide a balanced equation for the overall reaction to help answer this question below. Hint - track your boron: 3. (4pts) Provide the structure of the unknown ketone/aldehyde starting material that would produce the following HNMR spectrum: 8 2H, Doublet 2H, Doublet 6 5 2H, Quartet 4 PPM 3 2H, Triplet 3H, Triplet 2H, Complex multiplet resembling a sextet 2 1 3H, Triplet 4. (4pts) Students are asked to "wash" their reaction solution, as well as, the organic extraction layers. What is/are the most likely impurities that are removed during these washing steps? Provide a balanced equation for the overall reaction to help answer this question below. Hint - track your boron:
Expert Answer:

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
What are the observational units? To help answer this question Pew Research(2009) conducted a survey of nationally representative 242 cell phone users ages 16 to 17 years and asked whether they had...
-
For each of the following cases, provide the structure of an alkene that would give the alcohol as the major (or only) product of hydroboration--oxidation. CH CH CH CH OH CH, CH,
-
Students are asked to rate their preference for one of four video games. The following table lists the observed preferences in a sample of 120 students. State whether to reject or retain the null...
-
Elizabeth Is a nurse, and she just administered 1.8 milliliters of medication to one of her patients. Elizabeth knows that the amount of medication remaining in the patient's body wi decrease by a...
-
What formula do you use to calculate ROI?
-
Firewall Project XT. Using the complexity weighting scheme shown in Exercise and the function point complexity weighted table shown below, estimate the total function point count. Assume historical...
-
Purina entered in a contract with the defendant to sell the defendant piglets, known as weanlingsbaby pigs that have been weaned. It is uncontested that the buyer breached and that Purina is entitled...
-
The current assets and current liabilities sections of the balance sheet of Agincourt Company appear as follows. The following errors in the corporation's accounting have been discovered: 1. January...
-
2. (7pts) Give an example of a 44 matrix A which have the following properties: (a) A is lower triangular. All its entries are integers. (b) A has two distinct eigenvalues. Each eigenvalue is a digit...
-
A mass-transfer process is used to remove ammonia (NH 3 , solute A) from a mixture of NH 3 and air, using water as the absorption solvent. At the present conditions of operation, the partial pressure...
-
The shareholders of the Mango Company need to elect 10 new directors. There are 950,000 shares outstanding currently trading at $70 per share. You would like to serve on the board of directors;...
-
How to analyze decision scenario cases, executive summary.
-
1.0 8 10 If f f(x) dx = 17 and f(x)dx = 12, find r=12, find [(3(x)+2)dx 0 8
-
(a) Consider a two-period binomial setting. The current price of a stock is 80. For each of the next two periods, the stock will either move up by 10% or down by 10%. The risk-free rate per period is...
-
Imagine that you have a 500 g iron pot (c = 0.440 C), a 500 g copper pot (c = 0.385.C), and a 500 g J aluminium pot (c = 0.897 8 C). You fill each pot with 250 mL of water and heat the water to 100C...
-
A $10,000 bond with a 7% coupon rate is redeemable on September 1, 2024. What was its purchase price on October 18, 2013 when the yield rate was 9% compounded semi-annually? 0 Purchase Date:...
-
The Aggarwal Corporation needs to save $7 million to retire a $7 million mortgage that matures in 16 years. To retire this mortgage, the Company plans to put a fixed amount into an account at the end...
-
Rewrite the code of Figure 7.3 in Ada, Java, or C#. Figure 7.3: template class queue { item items [max_items]; int next_free, next_full, num_items; public: queue () : next_free (0), next_full(0),...
-
(a) The iSH group is sometimes called the mercapto group. 6-Mercaptopurine is used in the treatment of acute leukemia. Write its structure. (b) Allopurinol, a compound used to treat gout, is...
-
If benzene were 1,3,5-cyclohexatriene, the carbon-carbon bonds would be alternately long and short as indicated in the following structures. However, to consider the structures here as resonance...
-
When the bicyclic alkene I, a trans-decalin derivative, reacts with a peroxy acid, II is the major product. What factor favors the formation of II in preference to III? (You may find it helpful to...
-
Prove Part 1 of Theorem 1.14 using induction. That is, prove that for any non-negative integer \(k\), \[H_{k}(x)=\sum_{i=0}^{\lfloor k / 2floor}(-1)^{i} \frac{(2 i) !}{2^{i} i...
-
Use Theorem 1.13 (Taylor) to find fourth and fifth order polynomials that are approximations to the standard normal distribution function \(\Phi(x)\). Is there a difference between the...
-
Prove Part 2 of Theorem 1.14. That is, prove that for any non-negative integer \(k \geq 2\), \[H_{k}(x)=x H_{k-1}(x)-(k-1) H_{k-2}(x) .\] The simplest approach is to use Definition 1.6. Theorem 1.14....

Study smarter with the SolutionInn App


