Question: A student's laboratory notebook shows the following graphical entry, Obviously, the student has neglected to label which of the two superimposed curves belongs to
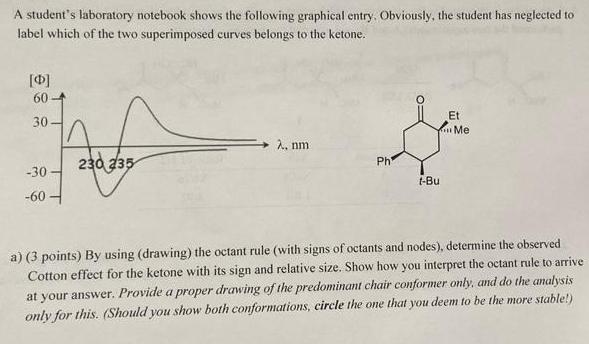
A student's laboratory notebook shows the following graphical entry, Obviously, the student has neglected to label which of the two superimposed curves belongs to the ketone. [0] 60 Et 30- Me A, nm 230 235 Ph -30 t-Bu -60 - a) (3 points) By using (drawing) the octant rule (with signs of octants and nodes), determine the observed Cotton effect for the ketone with its sign and relative size. Show how you interpret the octant rule to arrive at your answer. Provide a proper drawing of the predominant chair conformer only, and do the analysis only for this. (Should you show both conformations, circle the one that you deem to be the more stable!)
Step by Step Solution
3.42 Rating (152 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


