Two tanks are initially filled with 1 kL of pure water. A solution with 10 kg/kL of
Question:
Two tanks are initially filled with 1 kL of pure water. A solution with 10 kg/kL of salt is flowing into tank 1 at 5 kL/hr. A solution with 20 kg/kL of salt is flowing into tank 2 at 5 kL/hr. Both tanks are well mixed. The resulting solution is flowing from tank 1 into tank 2 at 3 kL/hr, and from tank 2 into tank 1 at 2 kL/hr. Tank 1 is being drained at 4 kL/hr and tank 2 is being drained at 6 kL/hr. Let x(t) be the amount of salt in tank 1 in kg, and y(t) be the amount of salt in tank 2 in kg.
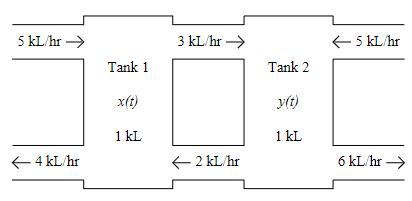
(a) Set up an IVP that models the amount of salt in each tank.
(b) Find the single equilibrium solution. Hint: Your equilibrium should have integer values for each component.
(c) On the next page, sketch a nullcline diagram for this system in the first quadrant. Include both the x and y-nullclines, with direction arrows on both nullclines, as well as in the large open regions between them. Finally, include two solutions curves: one that satisfies the initial data [0, 0]T and one that satisfies the initial data [40, 20]T . Once you’re satisfied, move onto parts (d) and (e) on the following page.
Carefully sketch your nullcline diagram for 1(c) here. You may use whatever scale you like, as long as you indicate it by labeling your axes.
Make sure your diagram includes all requested features, including:
• The x-nullcline(s).
• The y-nullcline(s).
• The equilibrium point from (b).
• Direction arrows on the nullclines.
• Direction arrows between the nullclines.
• The solutions requested: one satisfying [0, 0]T and one satisfying [40, 20]T .
• A scale of some sort.
(d) Based on your nullcline diagram, does the equilibrium you found in (a) appear to be stable, unstable, or semi-stable?
(e) Now, use the methods of Chapter 6 to find a solution to this IVP. Does your solution support your conclusion above? Why or why not?

Fundamentals Of Momentum Heat And Mass Transfer
ISBN: 9781118947463
6th Edition
Authors: James Welty, Gregory L. Rorrer, David G. Foster




