Question: 3. From the following vapor pressure data: a- Construct the Txy diagram using EXCEL at a total vapor pressure=1 atm, for the system benzene-toluene,
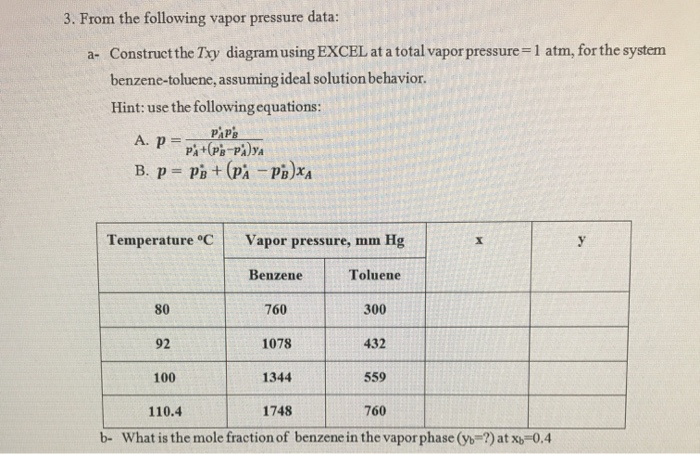
3. From the following vapor pressure data: a- Construct the Txy diagram using EXCEL at a total vapor pressure=1 atm, for the system benzene-toluene, assuming ideal solution behavior. Hint: use the following equations: PAP'S A. P= PA+ (PB-PAYA B. P= PB + (PA-PB)XA Temperature C 80 92 100 Vapor pressure, mm Hg Benzene 760 1078 1344 Toluene 300 432 559 X 110.4 1748 760 b- What is the mole fraction of benzene in the vapor phase (yb-?) at x -0.4 y
Step by Step Solution
There are 3 Steps involved in it
To solve the problem we need to construct a Txy diagram and calculate the mole fraction of benzene i... View full answer

Get step-by-step solutions from verified subject matter experts


