Draw on top of the free energy diagram below to show how changing the solvent from...
Fantastic news! We've Found the answer you've been seeking!
Question:
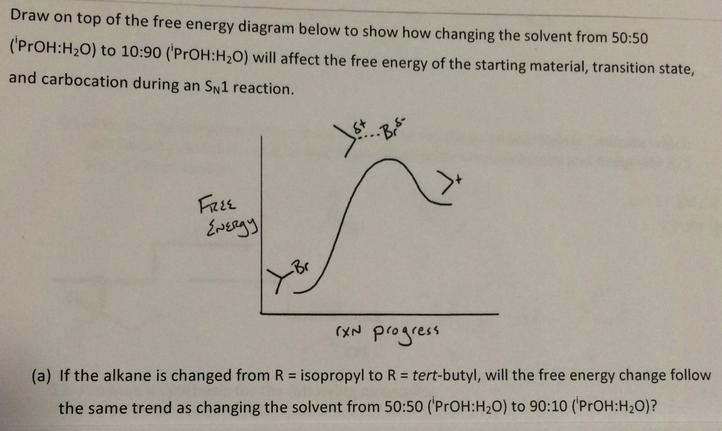
Transcribed Image Text:
Draw on top of the free energy diagram below to show how changing the solvent from 50:50 ('PrOH:H20) to 10:90 ('PrOH:H,0) will affect the free energy of the starting material, transition state, and carbocation during an SN1 reaction. FREE (XN progress (a) If the alkane is changed from R = isopropyl to R = tert-butyl, will the free energy change follow %3D the same trend as changing the solvent from 50:50 ('PrOH:H20) to 90:10 ('PrOH:H20)? Draw on top of the free energy diagram below to show how changing the solvent from 50:50 ('PrOH:H20) to 10:90 ('PrOH:H,0) will affect the free energy of the starting material, transition state, and carbocation during an SN1 reaction. FREE (XN progress (a) If the alkane is changed from R = isopropyl to R = tert-butyl, will the free energy change follow %3D the same trend as changing the solvent from 50:50 ('PrOH:H20) to 90:10 ('PrOH:H20)?
Expert Answer:
Answer rating: 100% (QA)
s hnging the slvent frm 5050 i rHH2 t 1090 i rHH2 ffet the free energy f ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
The following reaction involves a starting material with a double bond and a hydroxyl group, yet its mechanism resembles a pinacol rearrangement. Propose a mechanism, and point out the part of your...
-
The following tertiary alkyl bromides undergo an SN1 reaction in aqueous acetone to form the corresponding tertiary alcohols. List the alkyl bromides in order of decreasing reactivity. Br CH CCH3...
-
The free energy change for a reaction G is an extensive property. What is an extensive property? Surprisingly, one can calculate G from the cell potential for the reaction. This is surprising because...
-
A Moving to another question will save this response. Question 9 1 L The absent of the logic action Release in the Resource Module means: The operator will rework the entity a. b. C. The operator...
-
Policy Application: Tax Deductions and Tax Credits: In the U.S. income tax code, a number of expenditures are deductible. For most tax payers, the largest tax deduction comes from the portion of the...
-
Troy Engines, Ltd., manufactures a variety of engines for use in heavy equipment. The company has always produced all of the necessary parts for its engines, including all of the carburetors. An...
-
Give reasons for the following: (i) I.C. Engine cylinders are required to be cooled. (ii) Carburettor and spark plug are provided on petrol engines. The indicated power of a four stroke cycle petrol...
-
Develop a three-period moving average forecast for periods 1315. PERIOD DEMAND 10............ 248 11............ 370 12............ 424 13............ 286 14............ 444
-
Discuss comprehensively the following competencies and practice behaviors that are expected during your field practicum : Engage in ongoing self-reflective practice including an awareness of and...
-
Get It Right, CPAs, has been retained to review its client's corporate formation calculations for 20XX. Maria, Roger, and Novak created Grassroots Tennis, Inc. (GTI), which began operations on March...
-
1. One mole of a particular gas follows the state equation as given: P(V-B) = RT with B is a constant in unit atm . Derive equations to state the fugacity and fugacity coefficient of the gas. At 150...
-
Canadas real GDP was $1,830 billion in 2017 and $1,866 billion in 2018. Canadas population was 36.7 million in 2017 and 37.1 million in 2018. Calculate: a. The growth rate of real GDP. b. The growth...
-
The following figure shows the market for lows killed labor. The value of marginal product of high-skilled workers is $16 an hour greater than that of lows killed workers at each quantity of labor....
-
What are the rights and duties of sellers and buyers in a sales contract?
-
Explain how the pursuit of profit can sometimes lead to bank failures. U.S. bank profits rose during the first quarter of 2021, up $58.3 billion from a year ago. Loan balances decreased and deposits...
-
What happens if there is a shortage or a surplus of U.S. dollars in the foreign exchange market?
-
Bias in decision-making happens without thought in most decisions. Identify a recent current event in which you think bias was evident in the decision-making process surrounding that event. Briefly...
-
Some people argue that the internal control requirements of the Sarbanes-Oxley Act (SOX) put U.S. companies at a competitive disadvantage to companies outside the United States. Discuss the...
-
A chemist wants to synthesize 4-decyne but cannot find any 1-pentyne, the starting material used in the synthesis just described. How else can 4-decyne be synthesized?
-
According to the potential map for the ammonium ion, which atom(s) is (are) most positively charged?
-
Give the substitution products obtained when each of the following compounds is added to a solution of sodium acetate in acetic acid. a. 2-chloro-2-methyl-3-hexene b. 3-bromo-1-methylcyclohexene
-
What are the basic forms of international business activity?
-
Discuss the meaning of international business.
-
What is international business? How does it differ from domestic business?

Study smarter with the SolutionInn App


