3. Identify the chiral carbons in D-glucose D-glucose is shown here in its open chain form,...
Fantastic news! We've Found the answer you've been seeking!
Question:
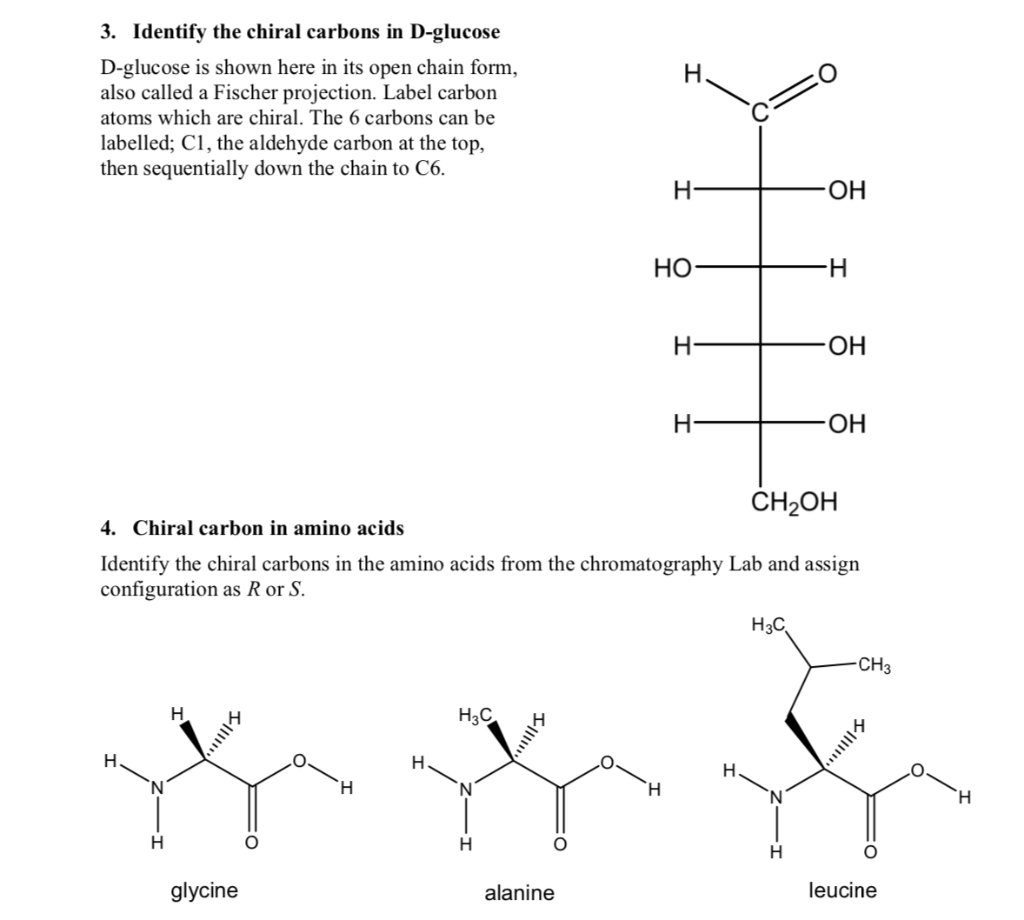
Transcribed Image Text:
3. Identify the chiral carbons in D-glucose D-glucose is shown here in its open chain form, also called a Fischer projection. Label carbon atoms which are chiral. The 6 carbons can be labelled; C1, the aldehyde carbon at the top, then sequentially down the chain to C6. H N H H I|||!!!! glycine H CH₂OH 4. Chiral carbon in amino acids Identify the chiral carbons in the amino acids from the chromatography Lab and assign configuration as R or S. H H₂C 112 alanine H- HO H H- H- H OH H3C -H OH OH -CH3 ||||| leucine 3. Identify the chiral carbons in D-glucose D-glucose is shown here in its open chain form, also called a Fischer projection. Label carbon atoms which are chiral. The 6 carbons can be labelled; C1, the aldehyde carbon at the top, then sequentially down the chain to C6. H N H H I|||!!!! glycine H CH₂OH 4. Chiral carbon in amino acids Identify the chiral carbons in the amino acids from the chromatography Lab and assign configuration as R or S. H H₂C 112 alanine H- HO H H- H- H OH H3C -H OH OH -CH3 ||||| leucine
Expert Answer:
Answer rating: 100% (QA)
Solution Concept Chival Carbon 3 D Glucose and to it 4 Gl... View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Posted Date:
Students also viewed these chemistry questions
-
Assign absolute configurations as R or S to each of the following compounds: a. b. c. H3C -CH2F CH3CH2 (+)-1-Fluoro-2-methylbutane CH3 C-CH2Br CH3CH2 (+)1-Bromo-2-methylbutane H3C -CH CH2 ...
-
Assign absolute configurations as R or S to each of the following compounds:
-
Circle the chiral carbons in the following compounds: O 0 H-C-H -C--CH, H-C-C HO-C-H H-C-H 00-
-
Using examples outline the main advantages and disadvantages of one of the following models of corporate governance: i) The principal/agent model ii) The stakeholder mode
-
An anticorrosive coating for a chemical storage tank will cost $5000 and last 5 years if touched up at the end of 3 years at a cost of $1000. If an oil-base enamel coating could be used that will...
-
Your boss is considering the use of a new predictor. The base rate is high, the selection ratio is low, and the validity coefficient is high for the current predictor. What would you advise your boss...
-
Titan Offroad Equipment uses the LIFO inventory method and values its inventory using the lower-of-cost-or-market^LCM) rule. Titan Offroad Equipment has the following account balances at December 31,...
-
Select a company you are interested in and obtain its annual reports by going to the companys website. Download the annual report for the most recent year. (On many companies websites, you will need...
-
4. Try again The answer is incorrect. 4 attempts remaining. Find the sum of the following geometric series: 7n-1 n=1 16" Please give the exact value of the sum, not an approximation. (Remember that...
-
I need help on this read from bottom to top. Image transcription text 7"} The second ?nancial statement to prepare is the statement of retained earnings. To determine the ending balance of...
-
A cylindrical log is cut as shown to form a park bench. Find: a. The area of triangle OAB in m. [2] b. The volume of the log in m. [3] B 70% 40 cm 3m
-
How do cells and organisms adapt their metabolic pathways in response to changing environmental conditions? Illustrate your answer with examples from cellular stress responses or shifts in nutrient...
-
A 5 3 2 nm pulse laser source produces a train of laser pulses with a 1 0 mJ energy at a 1 0 0 Hz repetition rate and a 1 0 nsec pulse width. How many photons are contained in each pulse? If the...
-
Walsh Company manufactures and sells one product. The following information pertains to each of the company's first two years of operations: Variable costs per unit: Manufacturing: Direct materials...
-
1. Characteristics of a quantitative study include all of the following except: a.clarifies underlying assumptions. b.asks who, what, why, where, when, or how. c.describes the relationship between...
-
1. The nurse wants to know how similar the 7-minute screen (7MS) is to the Mini-Mental State Examination (MMSE) in accurately screening for dementia. Can you distinguish the correct clinical question...
-
i need part B and the rest on excel Question 1: a. Prepare the amortization schedule for a thirty-year loan of $100,000. The APR is 3% and the loan calls for equal monthly payments. The following...
-
The relationship described in question 7 does not always appear to hold. What factors, besides the number of firms in the market, might affect margins?
-
In each of the following groups of three d aldoses, identify the two that give the same product upon Ruff degradation. (a) Galactose, gulose, talose; (b) Glucose, gulose, idose; (c) Allose, altrose,...
-
Ketoses show positive Fehlings and Tollenss tests not only by oxidation to -dicarbonyl compounds, but through a second process: Ketoses isomerize to aldoses in the presence of base. The aldose then...
-
Which of the following compounds are chiral? (a) 2-Methylheptane (b) 3-Methylheptane (c) 4-Methylheptane (d) 1,1-Dibromopropane (e) 1,2-Dibromopropane (f) 1,3-Dibromopropane (g) Ethene, H2C=CH2 (h)...
-
Brown & Brown, CPAs, was engaged by the board of directors of Cook Indus tries, Inc. to audit Cooks calendar year 19X5 financial statements. The following report was drafted by an audit assistant at...
-
Your client, S Company is a major manufacturer of widgets. Your audit report for the previous year was unqualified. During the current year, several class- action suits were filed against the company...
-
Q Company is a gas distribution company that is regulated by the state public service commission. The commission must approve all rate changes made by Q Company, but Q Company is allowed to collect...

Study smarter with the SolutionInn App


