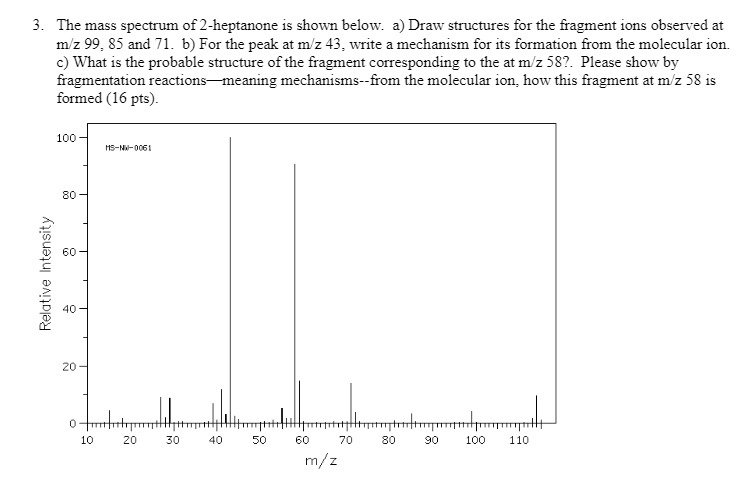
3. The mass spectrum of 2-heptanone is shown below. a) Draw structures for the fragment ions...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
3. The mass spectrum of 2-heptanone is shown below. a) Draw structures for the fragment ions observed at m/z 99, 85 and 71. b) For the peak at m/z 43, write a mechanism for its formation from the molecular ion. c) What is the probable structure of the fragment corresponding to the at m/z 587. Please show by fragmentation reactions meaning mechanisms--from the molecular ion, how this fragment at m/z 58 is formed (16 pts). 100 80 8 Relative Intensity 40 20 0 10 MS-NW-0061 20 30 40 50 60 70 m/z 80 Jump 90 100 110 3. The mass spectrum of 2-heptanone is shown below. a) Draw structures for the fragment ions observed at m/z 99, 85 and 71. b) For the peak at m/z 43, write a mechanism for its formation from the molecular ion. c) What is the probable structure of the fragment corresponding to the at m/z 587. Please show by fragmentation reactions meaning mechanisms--from the molecular ion, how this fragment at m/z 58 is formed (16 pts). 100 80 8 Relative Intensity 40 20 0 10 MS-NW-0061 20 30 40 50 60 70 m/z 80 Jump 90 100 110
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The molecular ion region in the mass spectrum of a large molecule, such as a protein, consists of a cluster of peaks differing by 1 Da. This pattern occurs because a molecule with many atoms has a...
-
10 20 30 40 50 60 70 Mancom 10 20 30 40 50 60 70 Macom B Ages of group of patients: The youngest participant has: Type of the graph: Ages of group of patients: The oldest participant has: Y/O Y/O ...
-
The mass spectrum of unknown compound A shows a molecular ion at m/z 116 and prominent peaks at m/z 87 and m/z 101. Its UV spectrum shows no maximum above 200 nm. The IR and NMR spectra of A follow....
-
Let B > 0. Show that the maximum of subject to the constraints x + this to conclude that f(x,...,xn) = X1 X2 Xn for all positive numbers a,..., an. . + xn = B and xj 0 for j = 1,. n occurs for x =...
-
A type of sampling application in which a relatively small initial sample is examined and decisions regarding expanding that sample are based on the results of this initial sample is known as a....
-
A counter flow, concentric tube heat exchanger used for engine cooling has been in service for an extended period of time. The heat transfer surface area of the exchanger is 5 m 2 , and the design...
-
Which of the following is an activity not usually associated with forensic accounting and fraud examination consulting and litigation support? 1. A. Assessing fraud risk associated with internal...
-
The Cutting Department of Cassel Company has the following production and cost data for July. Materials are entered at the beginning of the process. Conversion costs are incurred uniformly during the...
-
Would you make any changes to the Federal tax policy related to this area of the IRC ? If so , what would you change and why? How would these changes help rectify the disparate outcomes by race?
-
K Dhoni is a business consultant. The following trial balance is provided at 30 September 201: The following additional information is supplied: 1. At 30 September 201 rent received in advance...
-
You are an attorney in Florida that specializes in the preparation of Wills and Trusts. Based on your stellar reputation, Logan Roy, a billionaire, has hired you to create a will for him. You...
-
A gas station started the month with empty tanks, purchased 100,000 gallons at $2.00/gallon and ended with 10,000 gallons in the tanks at the end of the month. Calculate the Cost of Goods Sold for...
-
Exercise (1): The following data are some of the Comparative Statement of Profit or Loss of Salem Computin financial periods between 2015 and 2018, as Student Name Student ID ABCDEG s shown in figure...
-
Rojas Corporations comparative balance sheets are presented below. ROJAS CORPORATION Comparative Balance Sheets December 31 2020 2019 Cash $14,700 $11,000 Accounts receivable 21,100 23,800 Land...
-
ane, a partner in a CPA firm, wishes to borrow money from Company A, which her CPA firm audits. She is a covered member with respect to Company A. Which type of loan would be most likely to impair...
-
Case Study: Global Net Ltd is an internet service provider in Australia which employs staff locatedoverseas to man its call centres. It has been operating in the business for three years.Competition...
-
XXX Ltd. has banked with Bank of Australia for more than 10 years. XXX Ltd. would like to apply for a $ 10 million loan from the Bank of Australia and provides the following balance sheet report to...
-
Explain what is meant by vicarious liability and when it is available?
-
In Section 9-7B, we saw how acetylide ions add to carbonyl groups in much the same way as Grignard and organolithium reagents. Acetylide ions also add to ethylene oxide much like Grignard and...
-
Show the rest of the mechanism for formation of the cyclized intermediate in Figure 14-6. squalene epoxidase (enzyme) HC CH H CH HC CH squalene squalene-2.3-epoxide cyclized intermediate CH . CH H,C...
-
Predict the products of the following reactions. (a) Phenol + acetic anhydride (b) Phenol + acetic formic anhydride (c) Aniline + phthalic anhydride (d) Anisole + succinic anhydride and aluminum...
-
Is Fairmont in compliance with company policy that requires explicit approval of all hours of eighty hours or more.
-
d that Fairmont is not in complianThe forensic audit has determinece with Federal withholding requirements for FICA and Medicare because FICA and Medicare were not withheld from employee paychecks...
-
Which of the following is not a technique to conceal inventory shrinkage? 1. Counting and valuing the physical inventory at the end of each year 2. Writing off inventory after physical inventory...

Study smarter with the SolutionInn App



