3) XX has a new greenhouse project with an initial cost of $286,300 that is expected...
Fantastic news! We've Found the answer you've been seeking!
Question:
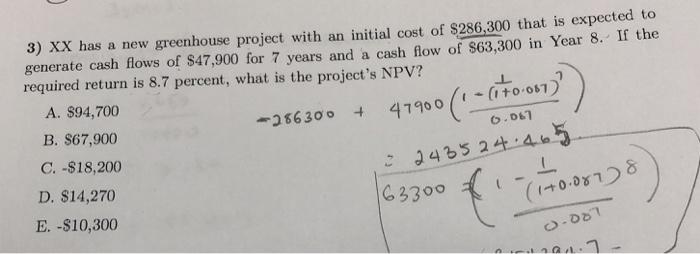
Transcribed Image Text:
3) XX has a new greenhouse project with an initial cost of $286,300 that is expected to generate cash flows of $47,900 for 7 years and a cash flow of $63,300 in Year 8. If the required return is 8.7 percent, what is the project's NPV? A. $94,700 B. $67,900 -286300 + 47900 (1 - (170-087) 0.067 C. -$18,200 22435 24.465 D. $14,270 E. -$10,300 163300 ( L (170.087)8) 0-087 3) XX has a new greenhouse project with an initial cost of $286,300 that is expected to generate cash flows of $47,900 for 7 years and a cash flow of $63,300 in Year 8. If the required return is 8.7 percent, what is the project's NPV? A. $94,700 B. $67,900 -286300 + 47900 (1 - (170-087) 0.067 C. -$18,200 22435 24.465 D. $14,270 E. -$10,300 163300 ( L (170.087)8) 0-087
Expert Answer:
Answer rating: 100% (QA)
Okay lets solve this stepbystep Initial cost of project 286... View the full answer

Related Book For 

Intermediate accounting
ISBN: 978-0077647094
7th edition
Authors: J. David Spiceland, James Sepe, Mark Nelson
Posted Date:
Students also viewed these accounting questions
-
Disco Kings is evaluating two mutually exclusive projects that have the following before-tax cash flow patterns: Project A Project B Initial outlay 200 000 350 000 Annual net cash flow 80 000 90 000...
-
You are considering an investment project with the cash flows of -300 (the initial cash flow), 800 (cash flow at year 1), -200 (cash flow at year 2). Given the discount rate of 10%, compute the...
-
Describe the companys top four customer segments. Incorporate descriptive statistics to your answer by identifying from each segment: (a) total revenue generated, (b) total number of transactions,...
-
Cubitene is a diterpene present in the defense secretion of a species of African termite. What unusual feature characterizes the joining of isoprene units in cubitene?
-
An alkyl halide reacts with an alkoxide ion to form a compound whose 1H NMR spectrum is shown here. Identify the alkyl halide and the alkoxide ion. 0 3 4 6 7 8 9 0
-
In Problems 5 44, solve each logarithmic equation. Express irrational solutions in exact form. log7 x = 3 log7 2
-
Indicate whether the actions listed a through \(h\), primarily represent initiatives for cost containment (CC), cost avoidance (CA), or cost reduction (CR). Indicate what costs were contained,...
-
You are considering an investment in the common stock of Keller Corp. The stock is expected to pay a dividend of $2 a share at the end of the year (D1 = $2.00). The stock has a beta equal to 0.9. The...
-
to meet the intersecting business needs of managing general agents and surplus lines, two professional organizations merged to form the
-
A pulpwood loader as shown in Figure P5-63 is to be designed to lift 2.5 kip. Select a steel and determine a suitable tubular cross section for the main upright member BF that has attachments for the...
-
Define sample, target population and sampling. In addition, discuss at least six sampling techniques
-
A restaurateur provides you with the following information for you to calculate the cost of debt and equity for her business venture: 1. She borrows $100,000 from a bank and agrees to pay the bank...
-
Gloria Corporation presents the following information for year 2020 & 2021 Sales Production Expenses Direct materials Direct labor Gross Profit Factory overhead Operating Expenses 2020 P 45,000 2021...
-
An enterprise produces and sells Item A. The selling price of this item, regardless of the size of the sales, is 20.00 per unit. Normal production and sales are 10,000 units per year. For 2012,...
-
1. Choose a crime involving a victim(s), a defendant and a court case. 2. Identify the Criminal Justice team i.e. law enforcement, prosecutor, victim advocate etc. 3. Research each person and write a...
-
A company's 2005 sales were $10 million. If sales grow at 8% per year, how large will they be in 8 years? 2. Suppose a government bond will pay $1,000 three years from now. If the going interest rate...
-
The EPS of Mass Energy Co. was $7.2 last year. It maintains a dividend payout ratio of 40%. Assume that the ROE of Mass Energy Co. is 16% and cost of equity is 13.5%. What price do you expect shares...
-
The landing gear of an aircraft with: mass of 2000 kg the spring-mass-damper system Consider that the runway surface is y(t) = 0.2 cos 157.08t stiffness of the spring is 5 x 105 N/m. What is the...
-
What criteria indicate that a debt investment should be accounted for at amortized cost?
-
At December 31, 2012, Cord Companys plant asset and accumulated depreciation and amortization accounts had balances as follows: Depreciation methods and useful lives: Buildings150% declining balance;...
-
During 2013, WMC Corporation discovered that its ending inventories reported on its financial statements were misstated by the following amounts: 2011 . understated by . $120,000 2012 . overstated by...
-
Monopolistic competition is similar to both _________ and perfect competition. As in monopoly, firms have some control over market _________, but as in perfect competition, they face _________ from...
-
Due to the free entry of new firms, long-run economic profits in monopolistic competition are _________.
-
Can advertising increase demand?

Study smarter with the SolutionInn App


