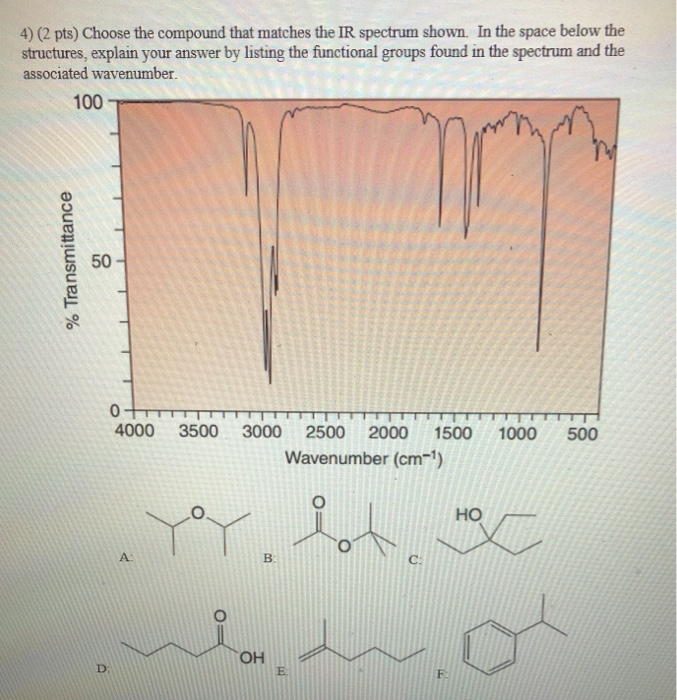
4) (2 pts) Choose the compound that matches the IR spectrum shown. In the space below...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
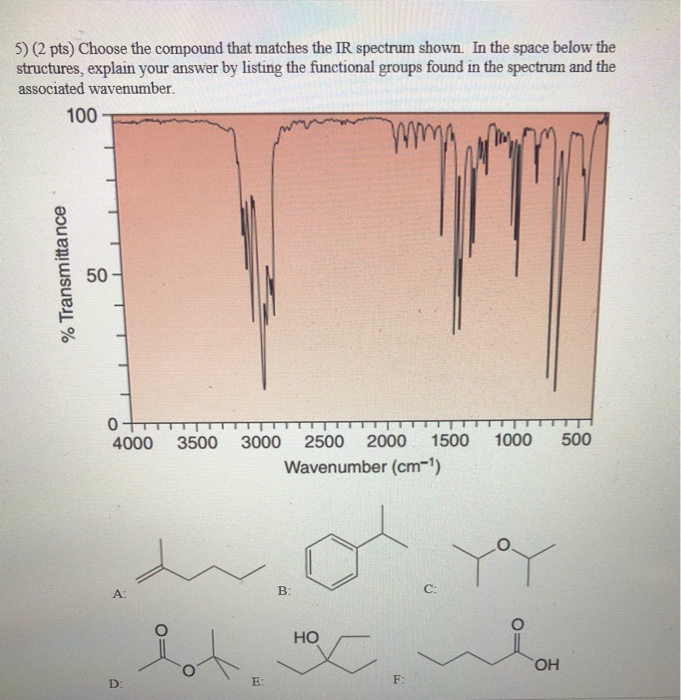
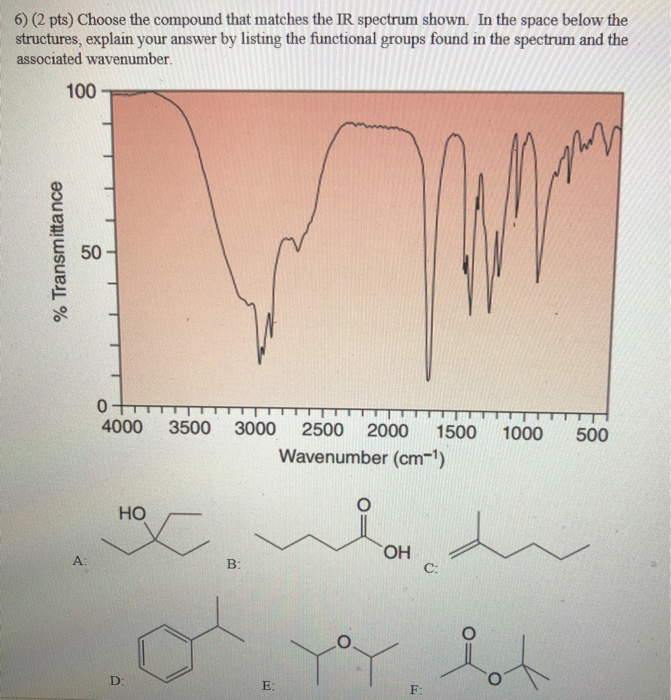
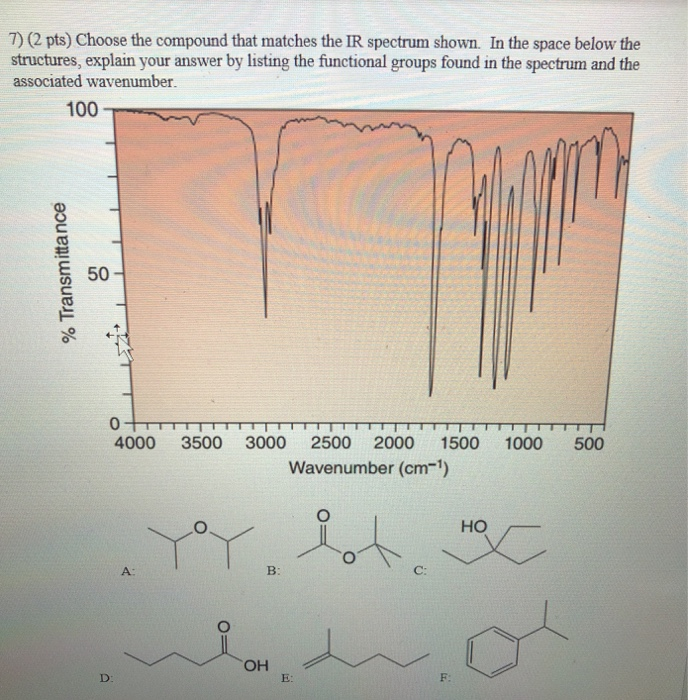
4) (2 pts) Choose the compound that matches the IR spectrum shown. In the space below the structures, explain your answer by listing the functional groups found in the spectrum and the associated wavenumber. % Transmittance 100 50- 0 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm-¹) D: A ya B: OH E: C: F HO M C 5) (2 pts) Choose the compound that matches the IR spectrum shown. In the space below the structures, explain your answer by listing the functional groups found in the spectrum and the associated wavenumber. 100 % Transmittance 50 0 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm-¹) A: D: E: B: HO F: C: YoY OH 6) (2 pts) Choose the compound that matches the IR spectrum shown. In the space below the structures, explain your answer by listing the functional groups found in the spectrum and the associated wavenumber. % Transmittance 100 50 A: 0 4000 3500 3000 2500 2000 1500 1000 Wavenumber (cm-¹) HO HOC D: B: E: la d OH C: F: Pot 500 7) (2 pts) Choose the compound that matches the IR spectrum shown. In the space below the structures, explain your answer by listing the functional groups found in the spectrum and the associated wavenumber. 100 % Transmittance 50 0 D: 4000 A: 3500 3000 2500 2000 1500 Wavenumber (cm-¹) Y B: OH E: C: F: 1000 HO HX 500 4) (2 pts) Choose the compound that matches the IR spectrum shown. In the space below the structures, explain your answer by listing the functional groups found in the spectrum and the associated wavenumber. % Transmittance 100 50- 0 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm-¹) D: A ya B: OH E: C: F HO M C 5) (2 pts) Choose the compound that matches the IR spectrum shown. In the space below the structures, explain your answer by listing the functional groups found in the spectrum and the associated wavenumber. 100 % Transmittance 50 0 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm-¹) A: D: E: B: HO F: C: YoY OH 6) (2 pts) Choose the compound that matches the IR spectrum shown. In the space below the structures, explain your answer by listing the functional groups found in the spectrum and the associated wavenumber. % Transmittance 100 50 A: 0 4000 3500 3000 2500 2000 1500 1000 Wavenumber (cm-¹) HO HOC D: B: E: la d OH C: F: Pot 500 7) (2 pts) Choose the compound that matches the IR spectrum shown. In the space below the structures, explain your answer by listing the functional groups found in the spectrum and the associated wavenumber. 100 % Transmittance 50 0 D: 4000 A: 3500 3000 2500 2000 1500 Wavenumber (cm-¹) Y B: OH E: C: F: 1000 HO HX 500
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate and check your answer by listing all the selections of size 2 that can be made from the letters a, b, c, d, e, and f. (9)
-
Compound A has molecular formula C 8 H 8 O. An IR spectrum of compound A exhibits a signal at 1680 cm -1 . The 1 H NMR spectrum of compound A exhibits a group of signals between 7.5 and 8 ppm (with a...
-
The IR spectrum of a compound with molecular formula C5H8O was obtained in CCl4 and is shown in Figure 13.42. Identify the compound. Wavelenga qum) 15 16 14 3600 340) 3800 3300 3000 280K 2600 2400...
-
The accounting records of Shinault Inc. show the following data for 2017 (its first year of operations). 1. Life insurance expense on officers was $9,000. 2. Equipment was acquired in early January...
-
1. Why is it important to define the project scope? How would you define the scope of the payroll project in this case? 2. Review each constraint and identify its characteristics: present vs. future,...
-
1. At the time of the case, what had Milkowski and the VoIP2.biz team not accomplished that several of the Board members felt should have been accomplished? 2. Why does Milkowski believe that...
-
On October 1, 2017, Gordon borrows \($150\),000 cash from a bank by signing a three-year installment note bearing 10% interest. The note requires equal payments of \($60\),316 each year on September...
-
The comparative statements of Beulah Company are presented below. Additional data: The common stock recently sold at $19.50 per share. Instructions Compute the following ratios for 2014. (a) Current....
-
Jane has been offered a research consultancy job to study the impact of inequality of poverty in five SADC countries of her choice. As her former classmate, Jane has asked you to help advise her on...
-
Allstar Inc. is considering a lockbox system that would reduce its float by three days. An expected 500 collections per day will be made to the lockbox with an average payment size of $1,000. The...
-
Why is it artificial to unitize fixed costs?
-
Construct a written research plan using sources to outline the goals of a legal research scenario. Demonstrate knowledge and application in the fundamentals of the research and writing process using...
-
Ben Hill contracted to purchase a house from Maria Altman. To prepare for the closing, a title search was conducted and the deed transferring the house to Ms. Altman was examined. The deed mentioned...
-
Read the following articles and then answer the questions below: . "8 Reasons that Explain the Importance of Job Satisfaction" https://www.marketing91.com/the-importance-of-job-satisfaction/ . "20...
-
In an experiment, 7.5mol of a gas are held in a 3.5L container at 4.5atm. What temperature (in C) does the gas exert if it is assumed to be ideal
-
(a) Let let and let be given by i) Is finjective? Briefly explain. ii) Is f surjective? Briefly explain. iii) Is f'bijective? Briefly explain. v) Let (b) Let iv) Does f have an inverse function?...
-
24) Luther Corporation Consolidated Income Statement Year ended December 31 (in Smillions) Total sales Cost of sales Gross profit Selling, general, and administrative expenses Research and...
-
a. Show that the expansion of q(x) in ascending powers of x can be approximated to 10 2x + Bx 2 + Cx 3 where B and C are constants to be found. b. Find the percentage error made in using the series...
-
Determine the structure of each of the following unknown compounds based on its molecular formula and its IR and 1H NMR spectra. a. C5H12O b. C6H12O2 c. C4H7ClO2 d. C4H8O2 26 27 28 29 14 1 6 5 8 0...
-
The second-order rate constant (in units of M-1 s-1) for acid-catalyzed hydration at 25°C is given for each of the following alkenes: a. Calculate the relative rates of hydration of the alkenes....
-
a. Which of the indicated bonds in each molecule is shorter? b. Indicate the hybridization of the C, O, and N atoms in each of the molecules. 1. 2. 3. 4. 5. CH3CCH2OH CH NH CH CH2N CHCH 3 CH3 CH 3
-
(a) Show for a 2D Hall bar of length \(L\) and width \(w\) that \(j=\sigma E\) (where \(j\) is the current density, \(E\) is the electric field, and \(\sigma\) is the conductivity) is equivalent to...
-
A European charity has an asset allocation at the beginning of the year consisting of the asset classes and weights shown in Exhibit 11. As Exhibit 11 reveals, the charity has a policy that the asset...
-
Wakuluk is most likely to make significant adjustments to her estimate of the future growth trend for which of the following countries? A. Country Y only B. Country Z only C. Countries Y and Z Neshie...

Study smarter with the SolutionInn App



