Question: 4. (20%) In Figure 3, a period loading function is illustrated, whose math- ematic form is T. Yu p(t) = po sin (wpt) 3
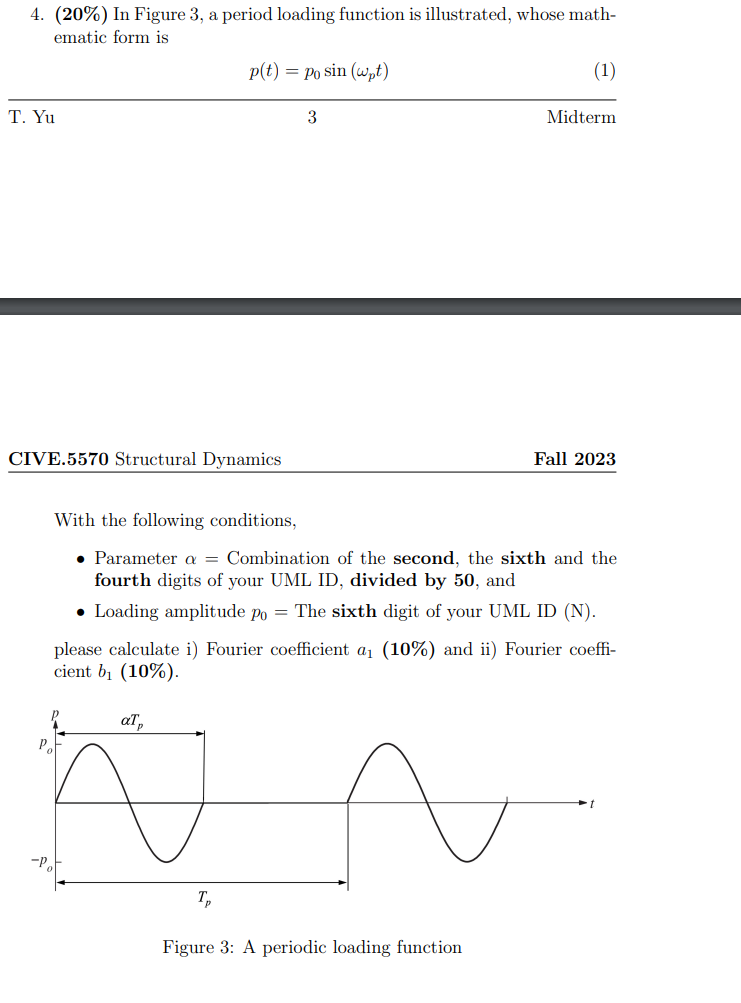
4. (20%) In Figure 3, a period loading function is illustrated, whose math- ematic form is T. Yu p(t) = po sin (wpt) 3 (1) Midterm CIVE.5570 Structural Dynamics With the following conditions, Fall 2023 Parameter a = Combination of the second, the sixth and the fourth digits of your UML ID, divided by 50, and Loading amplitude po = The sixth digit of your UML ID (N). please calculate i) Fourier coefficient a (10%) and ii) Fourier coeffi- cient b (10%). Figure 3: A periodic loading function
Step by Step Solution
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


