4. Absorber column design for solvent CO2 capture (25 points) A solvent absorption column is to...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
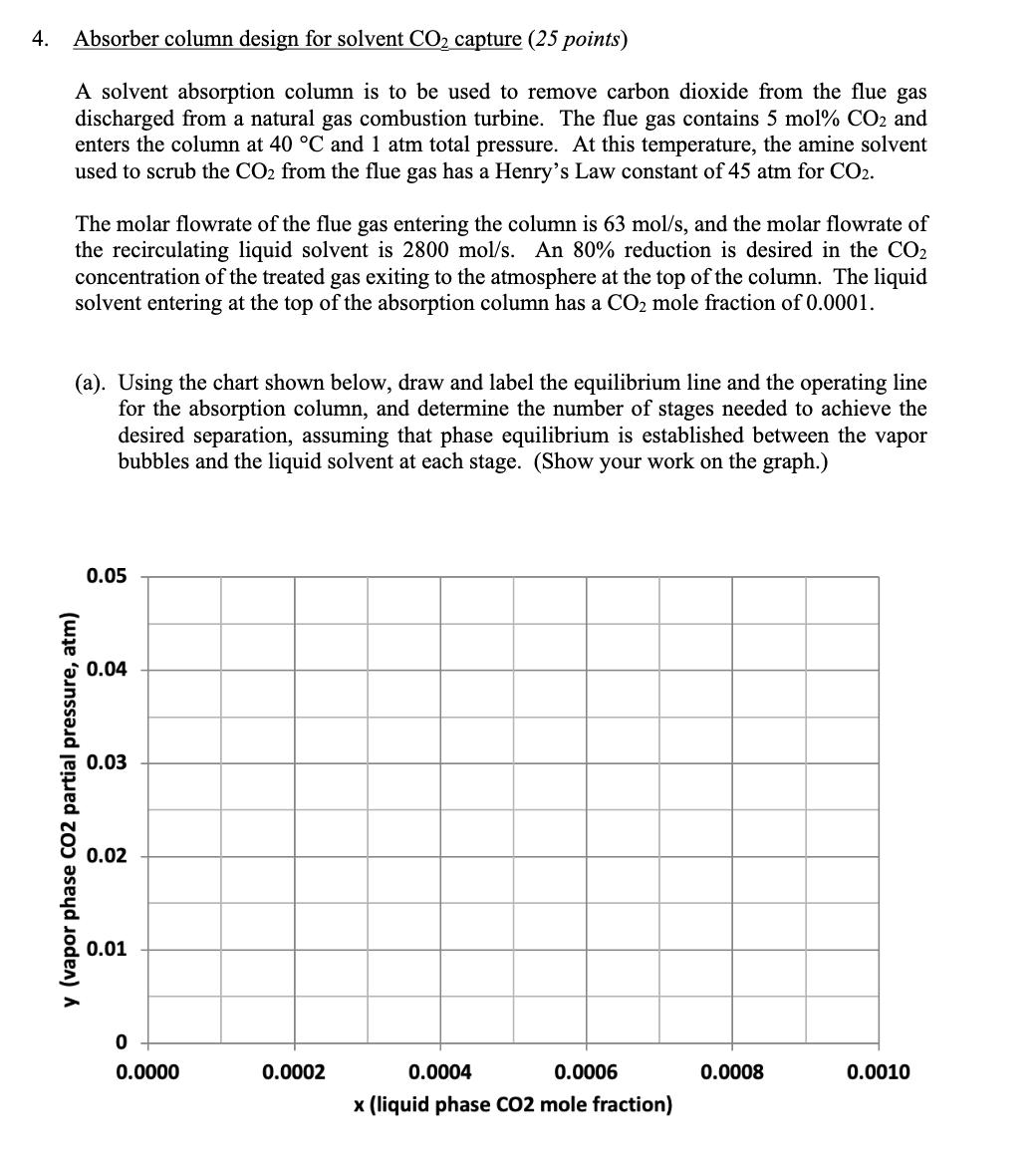
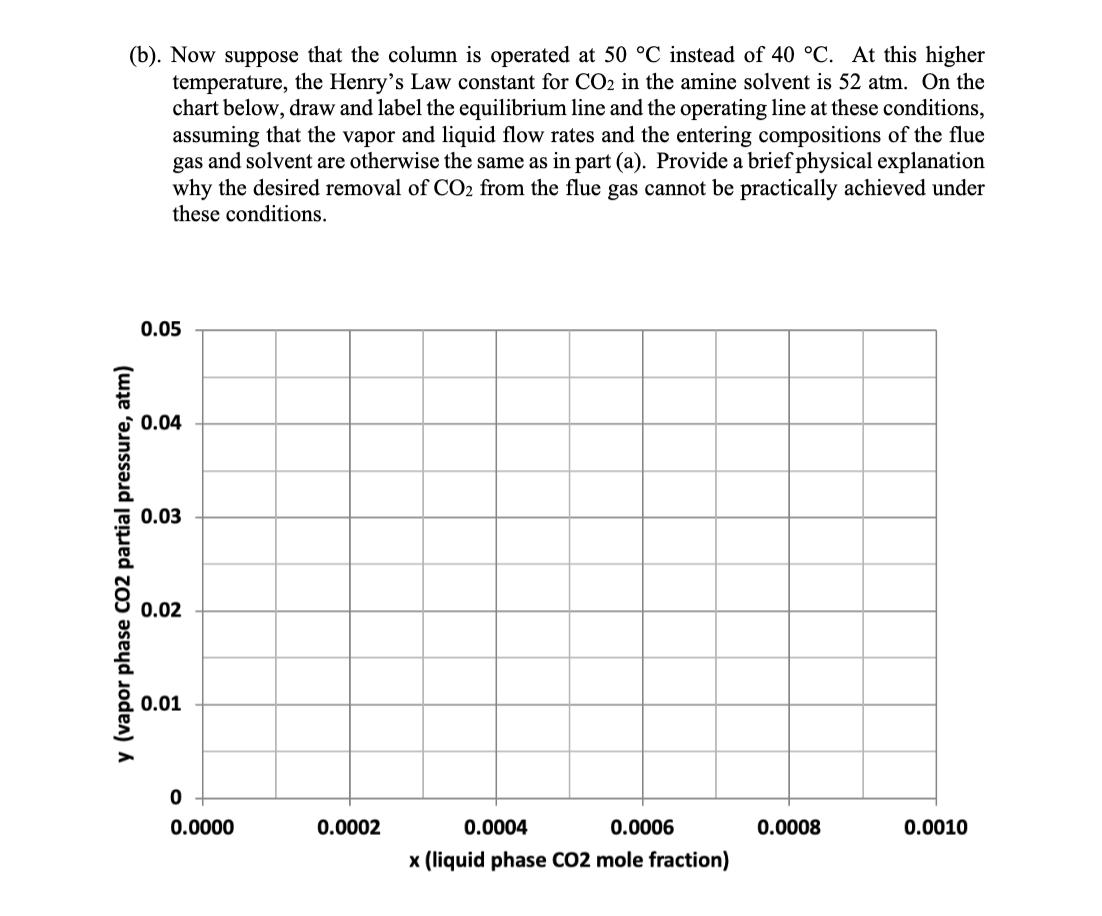
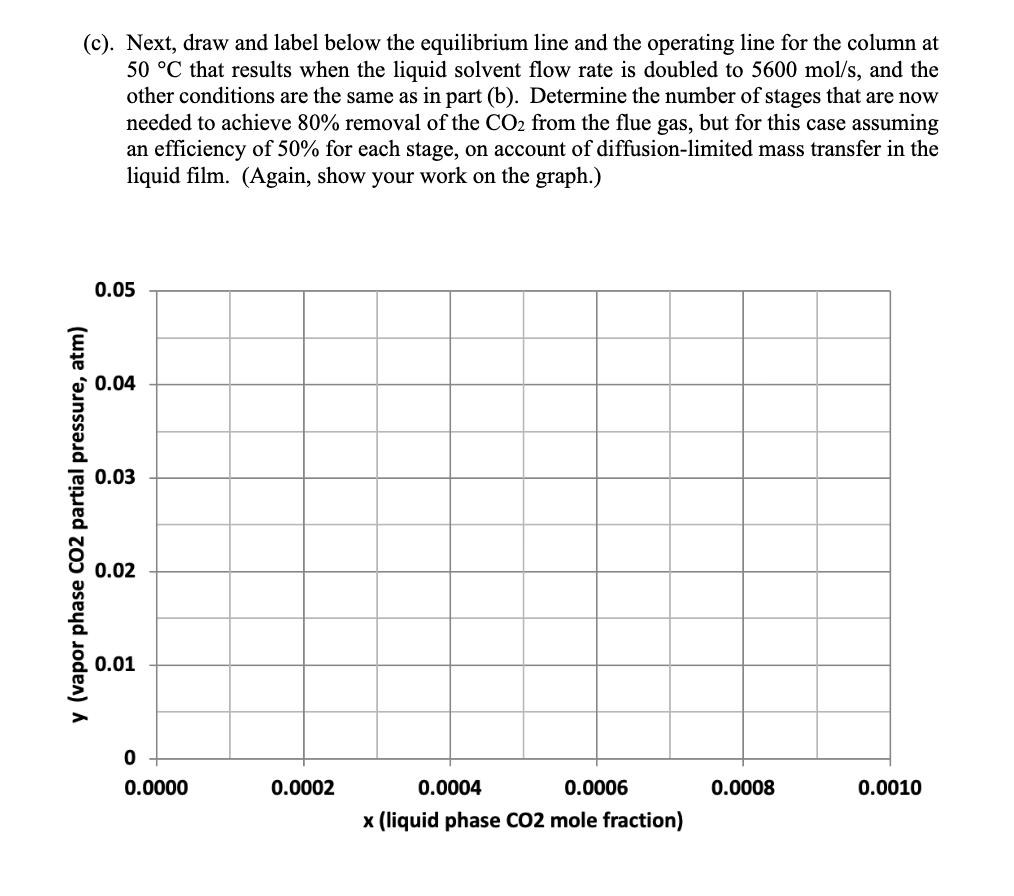
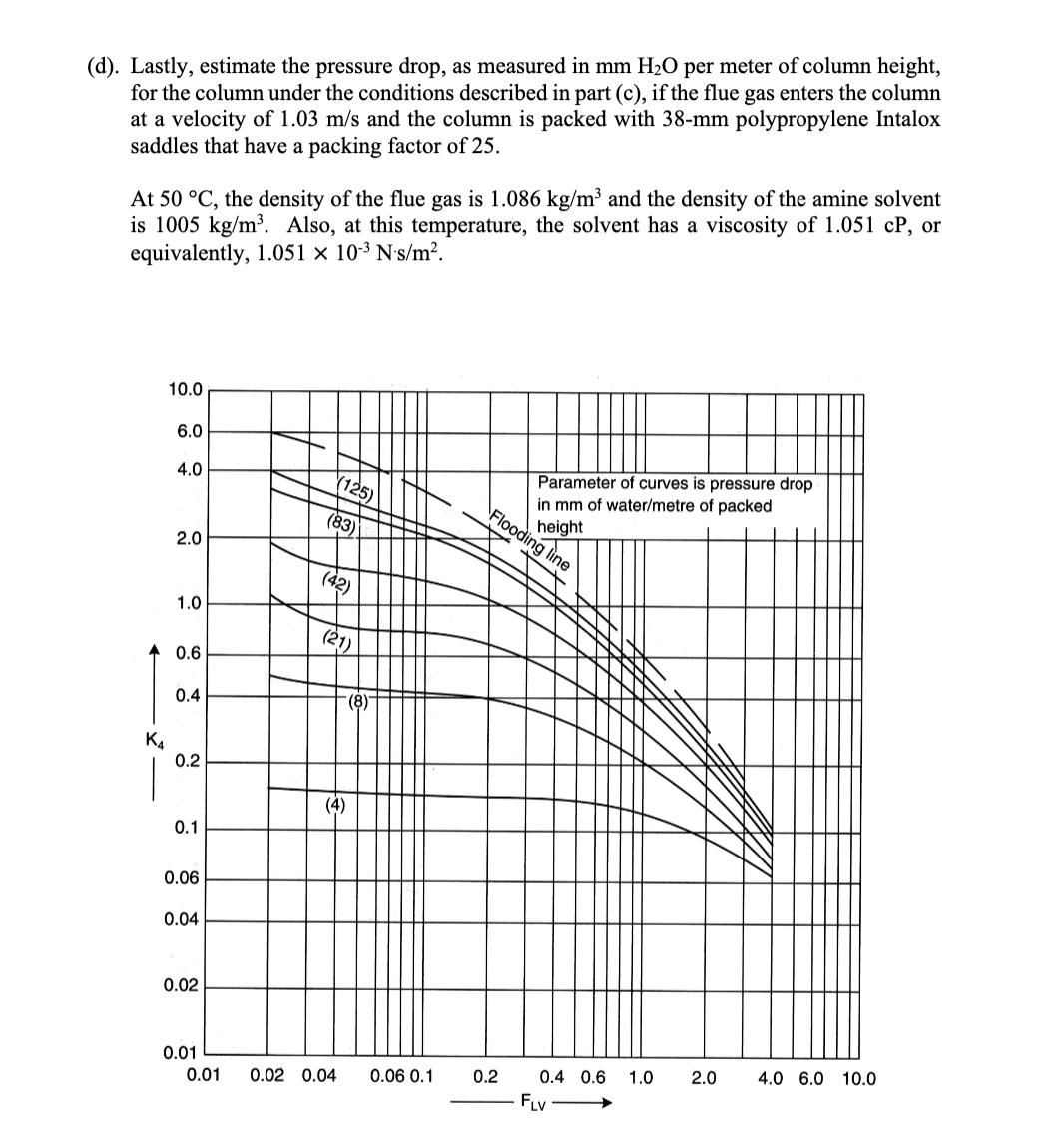
4. Absorber column design for solvent CO2 capture (25 points) A solvent absorption column is to be used to remove carbon dioxide from the flue gas discharged from a natural gas combustion turbine. The flue gas contains 5 mol% CO2 and enters the column at 40 C and 1 atm total pressure. At this temperature, the amine solvent used to scrub the CO2 from the flue gas has a Henry's Law constant of 45 atm for CO2. The molar flowrate of the flue gas entering the column is 63 mol/s, and the molar flowrate of the recirculating liquid solvent is 2800 mol/s. An 80% reduction is desired in the CO2 concentration of the treated gas exiting to the atmosphere at the top of the column. The liquid solvent entering at the top of the absorption column has a CO2 mole fraction of 0.0001. (a). Using the chart shown below, draw and label the equilibrium line and the operating line for the absorption column, and determine the number of stages needed to achieve the desired separation, assuming that phase equilibrium is established between the vapor bubbles and the liquid solvent at each stage. (Show your work on the graph.) 0.05 0.04 0.03 y (vapor phase CO2 partial pressure, atm) 0.02 0.01 0 0.0000 0.0002 0.0004 0.0006 0.0008 0.0010 x (liquid phase CO2 mole fraction) (b). Now suppose that the column is operated at 50 C instead of 40 C. At this higher temperature, the Henry's Law constant for CO2 in the amine solvent is 52 atm. On the chart below, draw and label the equilibrium line and the operating line at these conditions, assuming that the vapor and liquid flow rates and the entering compositions of the flue gas and solvent are otherwise the same as in part (a). Provide a brief physical explanation why the desired removal of CO2 from the flue gas cannot be practically achieved under these conditions. 0.05 0.04 0.03 y (vapor phase CO2 partial pressure, atm) 0.02 0.01 0 od 0.0000 0.0002 0.0004 0.0006 0.0008 0.0010 x (liquid phase CO2 mole fraction) (c). Next, draw and label below the equilibrium line and the operating line for the column at 50 C that results when the liquid solvent flow rate is doubled to 5600 mol/s, and the other conditions are the same as in part (b). Determine the number of stages that are now needed to achieve 80% removal of the CO2 from the flue gas, but for this case assuming an efficiency of 50% for each stage, on account of diffusion-limited mass transfer in the liquid film. (Again, show your work on the graph.) 0.05 0.04 0.03 y (vapor phase CO2 partial pressure, atm) 0.02 0.01 0 0.0000 0.0002 0.0004 0.0006 x (liquid phase CO2 mole fraction) 0.0008 0.0010 (d). Lastly, estimate the pressure drop, as measured in mm H2O per meter of column height, for the column under the conditions described in part (c), if the flue gas enters the column at a velocity of 1.03 m/s and the column is packed with 38-mm polypropylene Intalox saddles that have a packing factor of 25. At 50 C, the density of the flue gas is 1.086 kg/m and the density of the amine solvent is 1005 kg/m. Also, at this temperature, the solvent has a viscosity of 1.051 CP, or equivalently, 1.051 10-3 N's/m. 10.0 6.0 4.0 (125) (83) Flooding line 2.0 (42) 1.0 (21) 0.6 (8)- 0.4 K4 0.2 (4) 0.1 0.06 0.04 0.02 Parameter of curves is pressure drop in mm of water/metre of packed height 0.01 0.01 0.02 0.04 0.06 0.1 0.2 0.4 0.6 1.0 2.0 4.0 6.0 10.0 FLV 4. Absorber column design for solvent CO2 capture (25 points) A solvent absorption column is to be used to remove carbon dioxide from the flue gas discharged from a natural gas combustion turbine. The flue gas contains 5 mol% CO2 and enters the column at 40 C and 1 atm total pressure. At this temperature, the amine solvent used to scrub the CO2 from the flue gas has a Henry's Law constant of 45 atm for CO2. The molar flowrate of the flue gas entering the column is 63 mol/s, and the molar flowrate of the recirculating liquid solvent is 2800 mol/s. An 80% reduction is desired in the CO2 concentration of the treated gas exiting to the atmosphere at the top of the column. The liquid solvent entering at the top of the absorption column has a CO2 mole fraction of 0.0001. (a). Using the chart shown below, draw and label the equilibrium line and the operating line for the absorption column, and determine the number of stages needed to achieve the desired separation, assuming that phase equilibrium is established between the vapor bubbles and the liquid solvent at each stage. (Show your work on the graph.) 0.05 0.04 0.03 y (vapor phase CO2 partial pressure, atm) 0.02 0.01 0 0.0000 0.0002 0.0004 0.0006 0.0008 0.0010 x (liquid phase CO2 mole fraction) (b). Now suppose that the column is operated at 50 C instead of 40 C. At this higher temperature, the Henry's Law constant for CO2 in the amine solvent is 52 atm. On the chart below, draw and label the equilibrium line and the operating line at these conditions, assuming that the vapor and liquid flow rates and the entering compositions of the flue gas and solvent are otherwise the same as in part (a). Provide a brief physical explanation why the desired removal of CO2 from the flue gas cannot be practically achieved under these conditions. 0.05 0.04 0.03 y (vapor phase CO2 partial pressure, atm) 0.02 0.01 0 od 0.0000 0.0002 0.0004 0.0006 0.0008 0.0010 x (liquid phase CO2 mole fraction) (c). Next, draw and label below the equilibrium line and the operating line for the column at 50 C that results when the liquid solvent flow rate is doubled to 5600 mol/s, and the other conditions are the same as in part (b). Determine the number of stages that are now needed to achieve 80% removal of the CO2 from the flue gas, but for this case assuming an efficiency of 50% for each stage, on account of diffusion-limited mass transfer in the liquid film. (Again, show your work on the graph.) 0.05 0.04 0.03 y (vapor phase CO2 partial pressure, atm) 0.02 0.01 0 0.0000 0.0002 0.0004 0.0006 x (liquid phase CO2 mole fraction) 0.0008 0.0010 (d). Lastly, estimate the pressure drop, as measured in mm H2O per meter of column height, for the column under the conditions described in part (c), if the flue gas enters the column at a velocity of 1.03 m/s and the column is packed with 38-mm polypropylene Intalox saddles that have a packing factor of 25. At 50 C, the density of the flue gas is 1.086 kg/m and the density of the amine solvent is 1005 kg/m. Also, at this temperature, the solvent has a viscosity of 1.051 CP, or equivalently, 1.051 10-3 N's/m. 10.0 6.0 4.0 (125) (83) Flooding line 2.0 (42) 1.0 (21) 0.6 (8)- 0.4 K4 0.2 (4) 0.1 0.06 0.04 0.02 Parameter of curves is pressure drop in mm of water/metre of packed height 0.01 0.01 0.02 0.04 0.06 0.1 0.2 0.4 0.6 1.0 2.0 4.0 6.0 10.0 FLV
Expert Answer:

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Posted Date:
Students also viewed these chemical engineering questions
-
Design a process to remove carbon dioxide from the atmosphere that can be modularized for use on a community scale. Additionally, teams will consider the long-term storage of the sequestered CO2 and...
-
Consider a situation where the marginal damage function is known and equal to D'(E)= d.E. The aggregate marginal abatement cost curve is given by -C'(E)=-bE where is random variable uniformly...
-
Multiple Choice Questions 1. When using departmental overhead rates, which of the following cost objects is the first in the cost allocation process? a. Activities b. Units of product c. Product...
-
Subsequent payments testing is: (a) Vouching for a sample of payments made after year end to supporting invoices to ensure that amounts related to invoices dated prior to year end have been included...
-
What is the expectations gap?
-
For nearly 20 years Custom Coatings has provided painting and galvanizing services for manufacturers in its region. Manufacturers of various metal products have relied on the quality and quick...
-
Let's use this discussion to discuss any problems you are having with the material as you prepare for the final exam. We're also going to use the discussion to complete a required activity with your...
-
Davids Sport Supplies is a store that sells sports equipment and gear for teenagers and young adults. Davids is in the process of assigning the location of storage areas in its warehouse (Figure...
-
Days of Slumber sells mattress for cash and on credit. At the end of 2014, the following appeared in the companys balance sheet: Accounts receivable, net of $2,460 allowance$166,200 Which one of the...
-
Your bike, you start and accelerate to a speed of 5 m/s in 30 s over a distance of 75 m, and keep going at this speed on a level ground for a further 1 km. During your trip, the average drag force on...
-
The Schrdinger equation of a particle confined to the positive x-axis is: - dy 2m dx 2 and the trial function of: -+mgxy = Ey Y(x) = x -ax with Y(0) = 0, Y (x) 0 as x and E is the energy...
-
A movie stunt driver on a motorcycle speeds horizontally off a 50.0m high cliff. How fast must the motorcycle leave the cliff top to land on the level ground below, 90.0m from the base of the cliff...
-
A photon with wavelength equal to 800 nm (infrared) strikes an electron, resulting in a second photon that is scattered at an angle 45 with respect to the first. (a) Calculate the difference in the...
-
A paper airplane is given a push so that it could fly 7 m / s north, but there is wind that also pushes it 8 m / s east. Use the simulation to solve the problem. How fast could it go ?
-
Explain the industrial significance and the effectiveness of the extended surfaces (fins).
-
The figure shows six containers, each of which is filled from the top. Assume that water is poured into the containers at a constant rate and each container is filled in 10 seconds. Assume also that...
-
A liquid mixture containing 70.0 mol of n -pentane and 30.0 mol of n -hexane initially at 46?C is partially vaporized at P = 1 atm in a single-stage distillation apparatus (Rayleigh still). The heat...
-
The Cookenwythe Gas Company pumps propane gas to the nearby Noxious Chemicals, Inc., polypropylene production plant. The gas is metered at the noxious plant at 400m 3 /h at 4.7atm gauge and 30C. The...
-
Natural gas that contains methane, ethane, and propane is to be burned with humid air. The adiabatic flame temperature is to be calculated from specified values of the following quantities: ycH 4 ,...
-
Which of the following is not one of the five major categories of fraudulent disbursements? 1. Payroll schemes 2. Expense reimbursement schemes 3. Shell company schemes 4. Billing schemes
-
Assume that your data analytics examination related to employee records resulted in the following summary table for employees with annual salaries greater than $75,000: Assume that your data...
-
Why was Adelphia cable company formed?

Study smarter with the SolutionInn App


