4. If a solution were to turn litmus blue, which of the following substances would most...
Fantastic news! We've Found the answer you've been seeking!
Question:
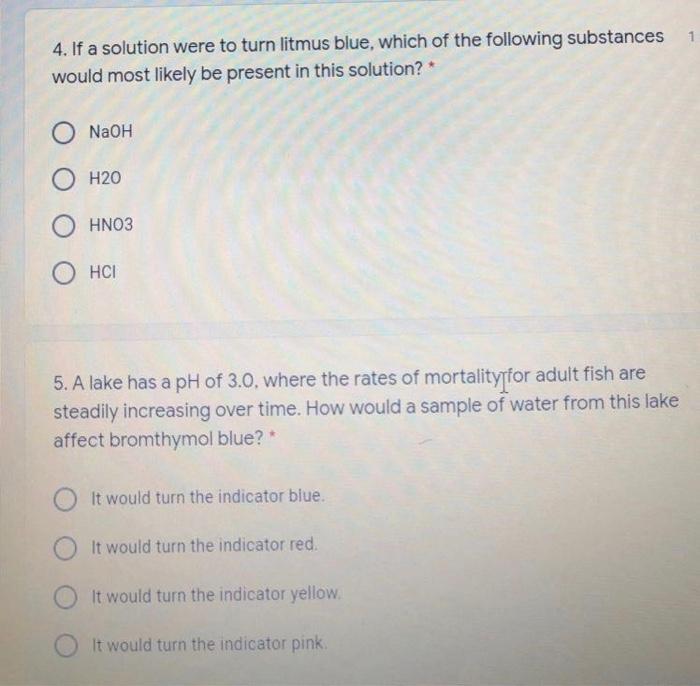
Transcribed Image Text:
4. If a solution were to turn litmus blue, which of the following substances would most likely be present in this solution? O NaOH H20 OHN03 HCI 5. A lake has a pH of 3.0, where the rates of mortality for adult fish are steadily increasing over time. How would a sample of water from this lake affect bromthymol blue? * OIt would turn the indicator blue. It would turn the indicator red. It would turn the indicator yellow. It would turn the indicator pink. 1 4. If a solution were to turn litmus blue, which of the following substances would most likely be present in this solution? O NaOH H20 OHN03 HCI 5. A lake has a pH of 3.0, where the rates of mortality for adult fish are steadily increasing over time. How would a sample of water from this lake affect bromthymol blue? * OIt would turn the indicator blue. It would turn the indicator red. It would turn the indicator yellow. It would turn the indicator pink. 1
Expert Answer:
Answer rating: 100% (QA)
Solution Answer 4 The correct answer is NaOH Explanation Litmus paper turns red for acid... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
Which of the following substances contain stereogenic centers? a. 2,2-dibromobutane b. 3-methylcyclopentene c. 1,2-difluoropropane d. 2,3-dimethylheptane e. methylcyclobutane f. 1-deuteriopropanol...
-
Which of the following activities would most likely be accounted for as a business-type activity? A. Fire protection. B. Recreation. C. Water operations. D. Street maintenance.
-
Which of the following accounts would most likely vary directly with the level of firm's sales? Discuss eachbriefly Ys No Cash Marketable Sec. Acct. Payable _Notes payable Plant and Equip Inventories
-
A company conducts a Security model workshop to review configuration options. The company uses Microsoft Entra ID to control access and permissions within multiple corporate applications. When users...
-
Fogelberg Corporation is a regional company, whose securities are thinly traded. Fogelberg has issued 10,000 units. Each unit consists of a $500 par, 12% subordinated debenture and 10 shares of $5...
-
SpongeFun Products manufactures and sells a variety of swimming products. Recently, the company opened a new plant to manufacture a lightweight, inflatable boat. Cost and sales data for 2016 are...
-
What is civil litigation and how does it differ from other types of litigation?
-
Lehmans net leverage ratio was not reported within the companys audited financial statements but rather in the companys financial highlights table and MD&A section of its annual report. What...
-
1. What are the challenges facing microfinance institutions in Myanmar? 2. Evaluate the credit risk management and mitigation used in Daung's current loan products? Case Study link -...
-
Get It Right, CPAs, has been retained to review its client's corporate formation calculations for 20XX. Maria, Roger, and Novak created Grassroots Tennis, Inc. (GTI), which began operations on March...
-
Arrange the following in order dH dehydrohalogenation :- (i) (iii) CI (iv) (1) (iii) > (iv) > (ii) > (i) (2) (iii) > (ii) > (i) > (iv) (3) (ii) > (iii) > (i) > (iv) (4) (i) > (ii) > (iii) > (iv) CI CI
-
Assume your clinic practice has the expenses outline in the Expense Grid table (fixed, semi-variable or variable) . You have been asked to perform an analysis of cost savings opportunities. In...
-
Two friends in a hurry to go picnicking decide tossed outside a window holding a cloth to catch a watermelon tossed out the window by a third friend Explain why a stretchy cloth is less likely to...
-
On January 1, 2XX1, Bargain Inc. acquired 100% of Wind Corporation for $9,750,000 cash. On that date, Willey's total stockholders' equity was $7,250,000. The following assets had fair values...
-
A parallel-plate capacitor with a 4.8 mm plate separation is charged to 65 V. What kinetic energy, in eV , must a proton launched from the negative plate be if it is just barely able to reach the...
-
Develop a simple set of financial statement that support your answers. TOM'S CLEANING COMPANY[1] Tom Willis is a 26-year-old ex-army man and a lifelong resident of Brockville, Ontario. Brockville is...
-
What is the weight of a body with 7.3 kg mass? Assume g=9.81 m/s 2 . Provide your answer in N and round to the nearest two decimal places.
-
Write a paper about medication error system 2016.
-
Calculate Kc at 303 K for SO2(g) + CI2(g) SO2CI2(g) if Kp = 34.5 at this temperature?
-
Consider the following reaction: 2 NO(g) + 2 H2(g) -- N2(g) + 2 H2O(g) (a) The rate law for this reaction is first order in H2 and second order in NO. Write the rate law. (b) If the rate constant for...
-
(a) Explain the difference between solubility and solubilityproduct constant. (b) Write the expression for the solubility-product constant for each of the following ionic compounds:MnCO3, Hg(OH)2,...
-
State which of the following items could appear as an asset on the statement of financial position of Goblin Combe plc, a leading premium drinks business: \($150,000\) of product sold during the...
-
Trading continues apace for your new business Climb On! The products have proved to be popular and, seeing this as your opportunity to seize the day, you decided to expand and grow the business. Here...
-
A rare book library at a University of England in England houses first edition (FE), original manuscript (OM), and authors journals (AJ). Some of these rare books are worth hundreds of thousands of...

Study smarter with the SolutionInn App


