4. The exit gas from an alcohol fermenter is an air-CO mixture containing 10 mol% CO...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
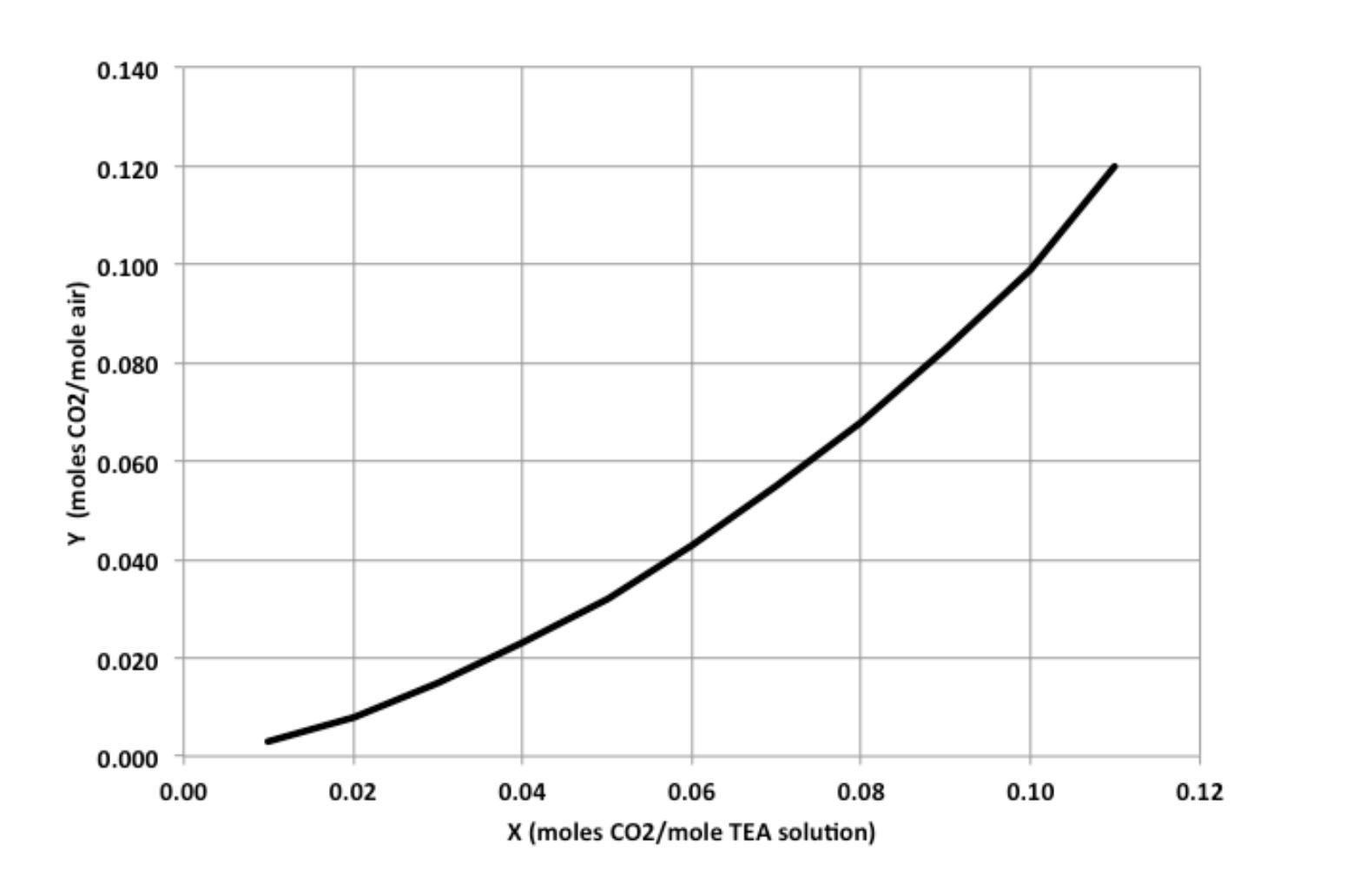
4. The exit gas from an alcohol fermenter is an air-CO mixture containing 10 mol% CO that is to be absorbed in a 5.0 M triethanolamine (TEA) solution containing 0.020 mol CO2/mol TEA. The column operates isothermally at 25 C, and the exit gas is to contain less than 2 mol% CO2. (a). If the tray column is operated at an absorbent flow rate 25% above the minimum required flow rate, but the stage efficiency is only 75% due to diffusion-limited mass transfer in the liquid film, how many trays are required to achieve the desired CO2 separation? (b). If the absorber operates without mass transfer resistance in the liquid film, but at an absorbent flow rate only 10% above the minimum required flow rate, how many trays are required to achieve the desired CO2 separation? (c). Suppose the absorber operates at 35% above the minimum required flow rate with no diffusion limitations, but the TEA solution entering the top of the tray column contains 0.025 mol CO2/mol TEA. How many trays are required in this scenario? Equilibrium data for Y vs. X at 25 C are shown in the plot on the next page, where Y = moles CO2/mole air and X = moles CO2/mole TEA solution. Y (moles CO2/mole air) 0.140 0.120 0.100 0.080 0.060 0.040 0.020 0.000 0.00 0.02 0.04 0.06 0.08 0.10 0.12 X (moles CO2/mole TEA solution) 4. The exit gas from an alcohol fermenter is an air-CO mixture containing 10 mol% CO that is to be absorbed in a 5.0 M triethanolamine (TEA) solution containing 0.020 mol CO2/mol TEA. The column operates isothermally at 25 C, and the exit gas is to contain less than 2 mol% CO2. (a). If the tray column is operated at an absorbent flow rate 25% above the minimum required flow rate, but the stage efficiency is only 75% due to diffusion-limited mass transfer in the liquid film, how many trays are required to achieve the desired CO2 separation? (b). If the absorber operates without mass transfer resistance in the liquid film, but at an absorbent flow rate only 10% above the minimum required flow rate, how many trays are required to achieve the desired CO2 separation? (c). Suppose the absorber operates at 35% above the minimum required flow rate with no diffusion limitations, but the TEA solution entering the top of the tray column contains 0.025 mol CO2/mol TEA. How many trays are required in this scenario? Equilibrium data for Y vs. X at 25 C are shown in the plot on the next page, where Y = moles CO2/mole air and X = moles CO2/mole TEA solution. Y (moles CO2/mole air) 0.140 0.120 0.100 0.080 0.060 0.040 0.020 0.000 0.00 0.02 0.04 0.06 0.08 0.10 0.12 X (moles CO2/mole TEA solution)
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The exit gas from an alcohol fermenter consists of an air-C02 mixture containing 10 mol% C02 that is to be absorbed in a 5.0-N solution of triethanolamine, containing 0.04 mol of carbon dioxide per...
-
What are the advantages for the HRM department if its head is on the main board? How might cultural influences noted in previous chapters affect that?
-
For nongovernmental NFP, how is the expiration of a time restriction recognized?
-
Let A(a) denote the area of the region bounded by y = e -ax and the x-axis on the interval [0, ]. Graph the function A(a), for 0 < a < . Describe how the area of the region decreases as the parameter...
-
What are the two categories of data mining and knowledge discovery software?
-
Special Revenue Fund, Voluntary Nonexchange Transactions. The City of Eldon applied for a competitive grant from the state government for park improvements such as upgrading hiking trails and bike...
-
c) Show that 2x+1 x (x+1) 1-2x2 = 2x + Hence, evaluate xx+1) S 2x+1 dx by first expressing the integrand as sum of partial fractions.
-
Honey Ltd, a New Zealand company, has sold US$150,000 of products to the US, to receive cash exactly one month later. At the time of sale, the spot rate of exchange is US$0.55, that is, NZ$1 buys...
-
The total payroll of Page Corporation was $4,320,000, of which $680,000 represented amounts paid in excess of $128,400 to certain employees. The amount paid to employees in excess of $7,000 was...
-
Consider the following address: 189.166.18.0/24. Give the: subnet mask, network, and broadcast addresses, for the first subnet upon borrowing 2 bit from the host portion to form subnets. O a....
-
Arobotics company is designing a swarm of drones for efficient parcel delivery in a city. Each drone is equipped with a sensor array that helps it avoid collisions with other drones. The challenge is...
-
Question 40 Assume that you define a function to delete a node in Doubly Linked List. Please select the appropriate code for the blank. public static Node deleteNode(Node deleted Note) { if (head...
-
Alice picks the two prime numbers p = 31, q = 47 as her secret numbers for an RSA cipher, and publishes the public key N, e with e = 11. a. What is N? b. Bob wishes to send an enciphered kiss to...
-
4. Given a signal as follows, compute the fundamental frequency, spectrum and bandwidth. Also calculate the channel capacity using Shanon criteria when SNR(db) = 250dB. s(t) = 5 sin(100nt) +...
-
Evaluate the function at the given value(s) of the independent variable. Simplify the results. (x) = cos 2x (a) (0) (b) (- /4) (c) (/3) (d) ()
-
Solve Exercise 10.11 using the UNIFAC method for Kvalues and obtain the converged solution.
-
One hundred pound-moles per hour of a mixture of 60 mol% methanol in water at 30oC and 1 atm is to be separated by distillation at the same pressure into a liquid distillate containing 98 mol%...
-
Air at 1 atm and 100C flows across a thin, flat plate of naphthalene that is 1 m long, causing the plate to sublime. The Reynolds number at the trailing edge of the plate is at the upper limit for a...
-
In the absence of limits on the behavior of large intermediaries, how might the perception of institutions being too-big-to-fail lead to increased concentration in the banking industry?
-
One goal of the regulatory reforms that followed the 2007-2009 financial crisis was to address the too-big-to-fail problem associated with large institutions. How did the reforms try to address this...
-
Analyze how a government regulates and supervises the financial system to contain risk.

Study smarter with the SolutionInn App


