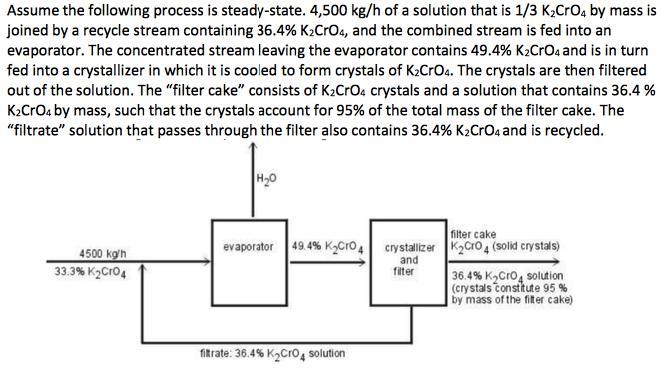
Assume the following process is steady-state. 4,500 kg/h of a solution that is 1/3 K2Cro, by...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Assume the following process is steady-state. 4,500 kg/h of a solution that is 1/3 K2Cro, by mass is joined by a recycle stream containing 36.4% K2CrO4, and the combined stream is fed into an evaporator. The concentrated stream leaving the evaporator contains 49.4% K2CrOa and is in turn fed into a crystallizer in which it is cooled to form crystals of K2CrOa. The crystals are then filtered out of the solution. The "filter cake" consists of K2CrOa crystals and a solution that contains 36.4 % K2CrOa by mass, such that the crystals account for 95% of the total mass of the filter cake. The "filtrate" solution that passes through the filter also contains 36.4% K2Croa and is recycled. H20 filter cake evaporator 49.4% KgCro, crystalizer K2cro4 (solid crystals) 4500 ko'h 33.3% K2Cro4 and fiter 36.4% K,Cro, solution (crystals constkute 95 % by mass of the fiter cake) fitrate: 36.4% K2Cro4 solution a. What are: (i) mass rate of evaporation, (ii) the mass rate of production of K2CrO4 crystals, (iii) the mass feed rates that the evaporator and the crystallizer must handle, and (iv) the recycle ratio? b. What would be the mass production rate of the crystals if the filtrate was discarded instead of recycled? Assume the following process is steady-state. 4,500 kg/h of a solution that is 1/3 K2Cro, by mass is joined by a recycle stream containing 36.4% K2CrO4, and the combined stream is fed into an evaporator. The concentrated stream leaving the evaporator contains 49.4% K2CrOa and is in turn fed into a crystallizer in which it is cooled to form crystals of K2CrOa. The crystals are then filtered out of the solution. The "filter cake" consists of K2CrOa crystals and a solution that contains 36.4 % K2CrOa by mass, such that the crystals account for 95% of the total mass of the filter cake. The "filtrate" solution that passes through the filter also contains 36.4% K2Croa and is recycled. H20 filter cake evaporator 49.4% KgCro, crystalizer K2cro4 (solid crystals) 4500 ko'h 33.3% K2Cro4 and fiter 36.4% K,Cro, solution (crystals constkute 95 % by mass of the fiter cake) fitrate: 36.4% K2Cro4 solution a. What are: (i) mass rate of evaporation, (ii) the mass rate of production of K2CrO4 crystals, (iii) the mass feed rates that the evaporator and the crystallizer must handle, and (iv) the recycle ratio? b. What would be the mass production rate of the crystals if the filtrate was discarded instead of recycled?
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A 250-mL portion of a solution that contains 1.5 mM Cu(NO 3 ) 2 is mixed with a solution that is 0.100 M NaCN. After equilibrium is reached, what concentration of Cu 2 + (aq) remains?
-
Calculate the ionic strength of a solution that is 0.040 mol kg-I in K3 [Fe (CN) 6J (aq), 0.030 mol kg-1 in KCI (aq), and 0.050 mol kg3 in NaBr (aq).
-
What is the pH of a solution that is 0.10 M KNO2 and 0.15 M HNO2 (nitrous acid)?
-
A strain of E-coli Beu 397-recA441 is placed into a nutrient broth at 30° Celsius and allowed to grow. The data shown in the table are collected. The population is measured in grams and the time...
-
What is the geometry about the metal center in this complex? Would you expect this complex to have counterions? Explain. N= M' N= N N=
-
What are the two methods of estimating your life insurance requirements?
-
Mass transfer from a bubble. Calculate the mass transfer coefficient for the air-water system for bubbles rising at a gas velocity of \(5 \mathrm{~cm} / \mathrm{s}\) in a pool of stagnant liquid. Use...
-
Little Switzerland Brewing Company was incorporated on January 28. On February 18, Ellison and Oxley were made directors of the company after they purchased some stock. Then, on September 25, Ellison...
-
The spot rate of interest is defined by st = 0.1 (0.8)t for t = 1, 2, 3, 4, 5. Find the present value of a 5-year annuity-due in which the first payment is equal to $1000, and each subsequent...
-
You want to park your bicycle in a bicycle parking area where bike racks are aligned in a row. There are already N bikes parked there (each bike is attached to exactly one rack, but a rack can have...
-
5.4 What will the following C code print on a little-endian machine like the x86? What will it print on a big-endian machine? unsigned short n = 0x1234; // 16 bits unsigned char *p (unsigned char *)...
-
How are annuities used as a retirement strategy?what would be a specific example.What are some disadvantages of using an annuity as a part of your strategy?
-
Frank's Fluids has a market value of Debt of $5,000,000 and outstanding Equity Valued at $50,000,000. The company has a WACC of 10%. The company's NOPAT is $7,500,000 and its EBITDA is $8,200,000....
-
What are some of the pros and cons of shifting elements of the portfolio, including rebalancing or making investment changes in terms of their tax liability? Showing full detail and specific examples...
-
Given the table below answer the question. 2011 2012 Cash Accounts receivable Inventory 200 100 800 1,400 3,600 3,100 Current assets 4,600 4,600 Gross fixed assets 22,000 31,000 (Accumulated...
-
Jack owns an initial wealth (W0) of $30. Inadvertently, he agreed to enter into the following game : - With 1/4 probability his wealth goes up to $50 - With 1/2 probability his wealth stays the same...
-
you are required to critically evaluate the key risk factors that led to the collapse of Silicon Valley Bank (SVB). SVB's collapse in March 2023 marked the second largest bank failure in U.S. history...
-
Show, if u(x, y) and v(x, y) are harmonic functions, that u + v must be a harmonic function but that uv need not be a harmonic function. Is e"e" a harmonic function?
-
Calculate the molar solubility of iron carbonate FeCO 3 in pure water.
-
Fluorine-18 is a radioactive isotope that emits positrons and is used in PET scans (positron emission tomography). It has a half-life of 110 minutes. If a patient is given a 1.5 ng sample of 18 F - ,...
-
Predict the silicate structure for the mineral talc Mg 3 Si 4 O 10 (OH) 2 that serves as the basis for talcum powder, and show that the formula is charge neutral.
-
What is the value of equity at time zero? A. 44,055. B. 77,973. C. 122,027. Mun Hoe Yip is valuing Pure Corporation. Pure is a simple corporation that is going out of business in five years,...
-
Economic income during Year 1 is closest to: A. 23,186. B. 29,287. C. 46,101. Mun Hoe Yip is valuing Pure Corporation. Pure is a simple corporation that is going out of business in five years,...
-
What is residual income during Year 1? A. 2,916. B. 2,542. C. 8,653. Mun Hoe Yip is valuing Pure Corporation. Pure is a simple corporation that is going out of business in five years, distributing...

Study smarter with the SolutionInn App


