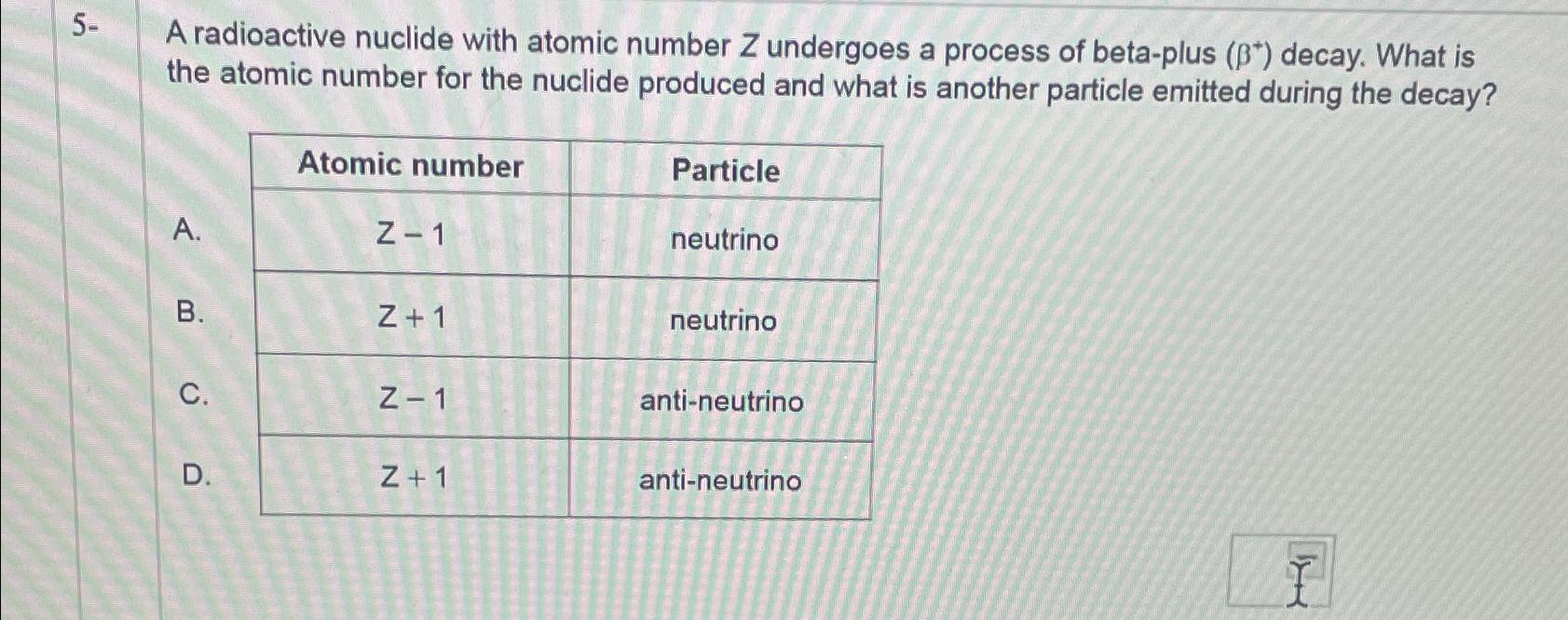
5- A radioactive nuclide with atomic number Z undergoes a process of beta-plus (*) decay. What...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
5- A radioactive nuclide with atomic number Z undergoes a process of beta-plus (ẞ*) decay. What is the atomic number for the nuclide produced and what is another particle emitted during the decay? Atomic number Particle A. Z-1 neutrino B. Z+1 neutrino C. Z-1 anti-neutrino D. Z+1 anti-neutrino 5- A radioactive nuclide with atomic number Z undergoes a process of beta-plus (ẞ*) decay. What is the atomic number for the nuclide produced and what is another particle emitted during the decay? Atomic number Particle A. Z-1 neutrino B. Z+1 neutrino C. Z-1 anti-neutrino D. Z+1 anti-neutrino
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below The image displays a ... View the full answer

Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Posted Date:
Students also viewed these physics questions
-
Use the contour map in Figure 20 to determine whether the critical points A, B,C, D are local minima, local maxima, or saddle points. y 2 0 -2. 1 A D 0 0 0 B 'C 23 2 x
-
When the 5-kg box reaches point A it has a speed v A = 10 m/s. Determine the normal force the box exerts on the surface when it reaches point B. Neglect friction and the size of the box. 9 m B - 9 m...
-
Which of the following graphs of a quadratic function has a negative y-intercept positive x-intercepts? A. B. C. -6 4 -2 -6 4 -2 -6 a 2 0 -2 y 4 0 2 y 4 B 4 -2 0 -2 2 4 6 6 X x X
-
On October 1, 2020, Berlin Corp. purchased 250, $ 1,000, 9% bonds for $ 260,000. An additional $ 7,500 was paid for the accrued interest, which is paid semi-annually on December 1 and June 1. The...
-
Briefly discuss about Employee Behavior and Organizational Design in the light of the following scenario. None of the factors that influence employee behavior exist in a vacuum. Spector (2013) says...
-
A steel wire is wrapped over a block of ice with two heavy weights attached to the ends of the wire. The wire passes through a block of ice without cutting the block in two. The ice melts under the...
-
1. To develop an understanding of your ethical leadership style 2. To understand how your preferred ethical leadership style relates to other ethical leadership styles Directions 1. Please read the...
-
This problem is based on the 2008 annual report of Intel Corporation in the appendix. Find in the Selected Financial Data (also known as the Five-Year Financial Summary), or calculate, the following...
-
Let n and m be positive integers with n m 1. (a) (9 points) How many different m-word sentences can be constructed using an al- phabet of n letters where each of the letters appears exactly once? A...
-
Using the below Adjusted Trial Balance, complete Reuben's Cubic Storage Income Statement. Statement of Changes to Owner's Equity and their Balance Sheet. When completing the Statement of Changes in...
-
What effect do lead-time and lot sizing have on an MRP? Demonstrate with an example of each.
-
considering I am new leader in FMCG company like TetraPak would like to driver Metrics and Evaluation: 1- How to Define key performance indicators (KPIs) to measure the success of the transformation?...
-
In order to prepare an audit on CSR and risk, perform some research on what should be audited in both CSR and risk. Find at least 3 outside sources for each and ensure they're listed in a references...
-
Has Knowledge Management come and gone? There was a time when Knowledge Management was the next big thing. Is it still? Having read many articles on beyond Knowledge Management, has Knowledge...
-
How can mindfulness, along with neuroscience/neuroleadership help a leader focus his or her followers? Please not that you do not have to cite the articles or include the references. It should...
-
For a MARR of 12%, which one of the two mutually exclusive machines should be selected? The increase in costs and benefits trend does not change when a new machine is put into action (The cost keep...
-
dx Q1. Evaluate the integral J:(**x}" using Trapezoidal Rule for n=4. Also find the value analytically and compare your result.
-
Portal Manufacturing has total fixed costs of $520,000. A unit of product sells for $15 and variable costs per unit are $11. a). Prepare a contribution margin income statement showing predicted net...
-
A lead electrode in 0.020 m Pb(NO 3 ) 2 (aq) is connected to a hydrogen electrode in which the pressure of H 2 is 1.0 bar. If the cell potential is 0.078 V at 25C, what is the pH of the electrolyte...
-
(a) Calculate the ratio of potential energies for an ionion interaction of Li + and of K + with the same anion. (b) Repeat the ratio calculation, but for an iondipole interaction between Li + and K +...
-
Explain why K + ions move more rapidly through water than Li + ions.
-
Find the modified \(z\)-transform of the following functions: (a) \(E(s)=\frac{6}{(s+1)(s+2)(s+3)}\) (b) \(E(s)=\frac{4}{s(s+2)^{2}}\) (c) \(E(s)=\frac{s^{2}+2 s+2}{s(s+2)^{2}}\) (d)...
-
Repeat Problem 4.8-1 after replacing the digital filter with this transfer function: \[ D(z)=\frac{z-0.5}{(z-0.8)(z-1)} \]
-
Consider the system of Fig. P4.10-1. The plant is described by the first-order differential equation \[ \frac{d y(t)}{d t}+0.04 y(t)=0.2 m(t) \] Let \(T=2 \mathrm{~s}\). (a) Find the system transfer...

Study smarter with the SolutionInn App


