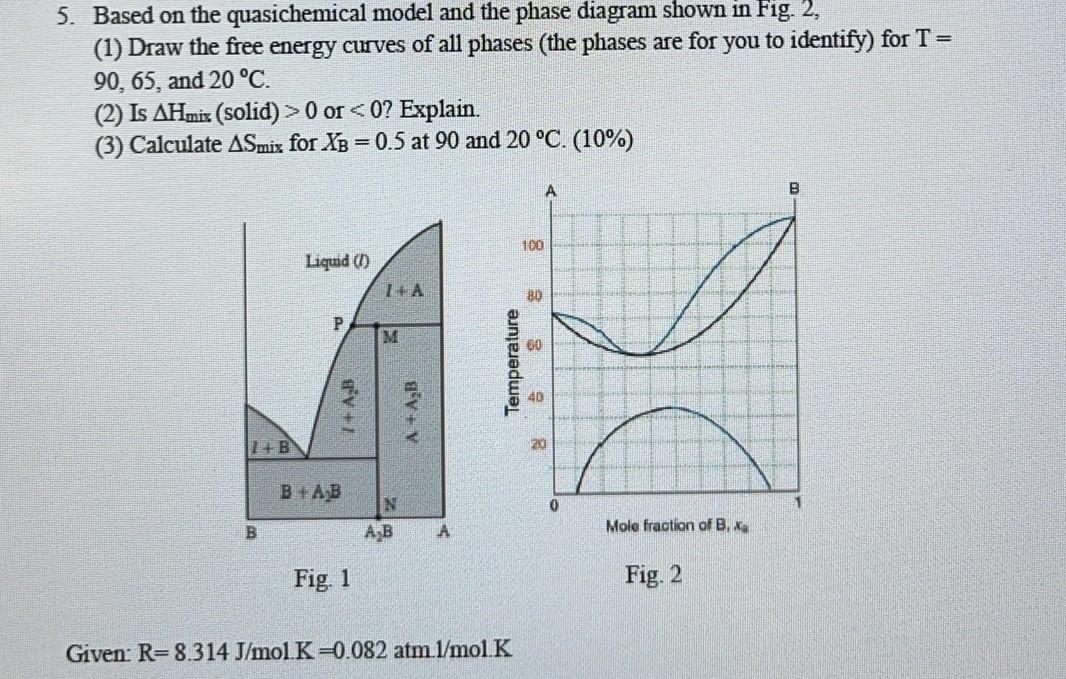
5. Based on the quasichemical model and the phase diagram shown in Fig. 2, (1) Draw...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
5. Based on the quasichemical model and the phase diagram shown in Fig. 2, (1) Draw the free energy curves of all phases (the phases are for you to identify) for T = 90, 65, and 20 °C. (2) Is AHmix (solid) >0 or <0? Explain. (3) Calculate ASmix for XB = 0.5 at 90 and 20 °C. (10%) B 100 Liquid () 1+A 60 40 14B 20 B+AB N. AB Mole fraction of B, x Fig. 1 Fig. 2 Given: R=8.314 J/mol K-0.082 atm.1/mol K 1+ AB A+A,B Temperature 5. Based on the quasichemical model and the phase diagram shown in Fig. 2, (1) Draw the free energy curves of all phases (the phases are for you to identify) for T = 90, 65, and 20 °C. (2) Is AHmix (solid) >0 or <0? Explain. (3) Calculate ASmix for XB = 0.5 at 90 and 20 °C. (10%) B 100 Liquid () 1+A 60 40 14B 20 B+AB N. AB Mole fraction of B, x Fig. 1 Fig. 2 Given: R=8.314 J/mol K-0.082 atm.1/mol K 1+ AB A+A,B Temperature
Expert Answer:
Answer rating: 100% (QA)
a At 90C it is in vapour phase Region I It is Homogeneous ... View the full answer

Related Book For 

Essentials of Materials Science and Engineering
ISBN: 978-1111576851
3rd edition
Authors: Donald R. Askeland, Wendelin J. Wright
Posted Date:
Students also viewed these chemistry questions
-
Draw a possible Feynman diagram using quarks (as in Fig 32-13c) for the reaction + p 0 + n.
-
The copper-silver phase diagram is shown in Figure 11-30. Copper has a higher melting point than silver. Figure 11-35 A phase diagram for elements A and B (for Problem 11-36). (a) Is copper element A...
-
The phase diagram for SO2 is shown here. (a) What does this diagram tell you about the enthalpy change in the reaction SO2(I) SO2(g)? (b) Calculate the equilibrium constant for this reaction at 100...
-
A politician claims that the mean salary for managers in his state is more than the national mean, $85,000. Assume the the population is normally distributed and the population standard deviation is...
-
Show that the variance is minimized for a fixed cost with the cost function in (3.12) when nh NhSh/ch, as in (3.13).
-
A sportswriter wants to know how strongly the residents of Indianapolis, Indiana, support the local minor league baseball team, the Indianapolis Indians. He stands outside the stadium before a game...
-
The nuclear reaction that powers the sun is the fusion of four protons into a helium nucleus. The process involves several steps, but the net reaction is simply \(4 \mathrm{p} ightarrow{ }^{4}...
-
Give examples of needs, wants, and demands that JetBlue customers demonstrate, differentiating these three concepts. What are the implications of each for JetBlues practices? In 2007, JetBlue was a...
-
Explain how a deviation from a cash-flow budget could lead to larger interest charges for a nonrevolving line of credit, even if the loan was originally designed at the beginning of the year to be...
-
Kat Ltd.s September balance sheet contains the following information: Cash.............................................................................$ 37,500 (dr) Accounts...
-
Using only the information provided in Exhibit 1, explain why further analysis of physician visits maybe needed. Compare the profitability of hospital and surgical services to physician services,...
-
reference Arranged Arranged (201 and be sure to explain and defend your respons 06..05 points, At least 10 words > Which character in Arranged (2017), (assigned directly above, quick link here...
-
Accounting for management decisions can be different from accounting to provide financial statements. One aspect of accounting for management decisions is the budgeting process. Describe the...
-
A 36-KN load P is supported by the three rods below. Determine the following: a. The change in length of rod EF. b. The stress in each rod. AAB = ACD = 200 mm AEF = 625 mm EST = 200 GPa A C P B FL E...
-
Subtract and simplify. 3 1 31 55 (Type a whole number or a simplified fraction) 5 5
-
Maya owns 3 different rings. She wears all 3 rings, but no 2 rings on the same finger, and none on her thumbs. Assuming she can wear any right on any finger, how many ways can she wear the 3 rings?...
-
Current Attempt in Progress Fill in the missing amounts for items in the following perpetual inventory records. (a) Using weighted average. (Round the weighted average cost per unit and total amounts...
-
What do you think?
-
How many grams of aluminum, with a dislocation density of 1010 cm/cm3, are required to give a total dislocation length that would stretch from New York City to Los Angeles (3000 miles)?
-
Figure 11-32 shows a cooling curve for an Al-Si alloy and Figure 11-19 shows the binary phase diagram for this system. Determine (a) The pouring temperature; (b) The superheat; (c) The liquidus...
-
Boron has a much lower coefficient of thermal expansion than aluminum, even though both are in the 3B column of the periodic table. Explain, based on binding energy, atomic size, and the energy well,...
-
Explain the steps in forming a limited liability corporation.
-
This chapter describes the common corporate forms of business organization in the United States. Business structures outside the United States, however, are often very different. Use the Internet to...
-
Midcore and NCT, with NCT being the surviving corporation. It subsequently filed for bankruptcy. Shareholders of the twocompanies sued, claiming that the officers and directors did not pay them as...

Study smarter with the SolutionInn App


