(5) Calculate the volume of CO gas at STP obtained by reacting a 10.00 g sample...
Fantastic news! We've Found the answer you've been seeking!
Question:
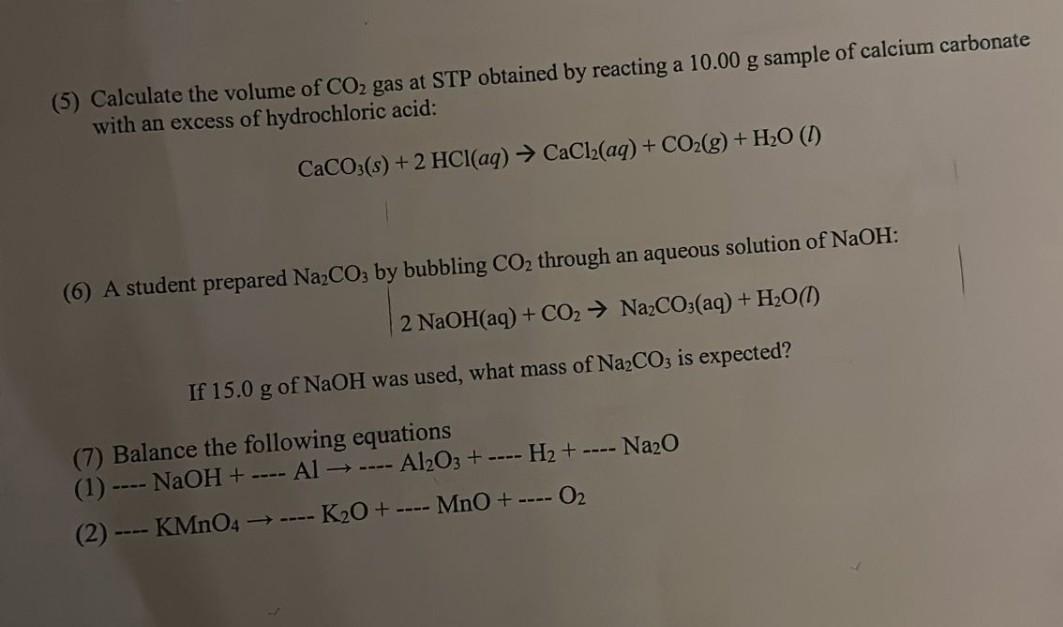
Transcribed Image Text:
(5) Calculate the volume of CO₂ gas at STP obtained by reacting a 10.00 g sample of calcium carbonate with an excess of hydrochloric acid: CaCO3(s) + 2 HCl(aq) → CaCl₂(aq) + CO₂(g) + H₂O (1) (6) A student prepared Na₂CO3 by bubbling CO₂ through an aqueous solution of NaOH: 2 NaOH(aq) + CO₂ → Na₂CO3(aq) + H₂O(1) If 15.0 g of NaOH was used, what mass of Na₂CO3 is expected? (7) Balance the following equations (1) - - NaOH + ---- (2) ---- KMnO4 → ---- ---- Al → ---- Al2O3+ - K₂O + H₂+ ---- MnO + ---- 0₂ Na₂O (5) Calculate the volume of CO₂ gas at STP obtained by reacting a 10.00 g sample of calcium carbonate with an excess of hydrochloric acid: CaCO3(s) + 2 HCl(aq) → CaCl₂(aq) + CO₂(g) + H₂O (1) (6) A student prepared Na₂CO3 by bubbling CO₂ through an aqueous solution of NaOH: 2 NaOH(aq) + CO₂ → Na₂CO3(aq) + H₂O(1) If 15.0 g of NaOH was used, what mass of Na₂CO3 is expected? (7) Balance the following equations (1) - - NaOH + ---- (2) ---- KMnO4 → ---- ---- Al → ---- Al2O3+ - K₂O + H₂+ ---- MnO + ---- 0₂ Na₂O (5) Calculate the volume of CO₂ gas at STP obtained by reacting a 10.00 g sample of calcium carbonate with an excess of hydrochloric acid: CaCO3(s) + 2 HCl(aq) → CaCl₂(aq) + CO₂(g) + H₂O (1) (6) A student prepared Na₂CO3 by bubbling CO₂ through an aqueous solution of NaOH: 2 NaOH(aq) + CO₂ → Na₂CO3(aq) + H₂O(1) If 15.0 g of NaOH was used, what mass of Na₂CO3 is expected? (7) Balance the following equations (1) - - NaOH + ---- (2) ---- KMnO4 → ---- ---- Al → ---- Al2O3+ - K₂O + H₂+ ---- MnO + ---- 0₂ Na₂O
Expert Answer:
Answer rating: 100% (QA)
The question youve presented is question 6 which requires calculating the expected mass of sodium ca... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
1. What mass of H2 should be produced by the reaction of Al with 75.0 mL of 2.95M HCl? 2Al(s) + 6HCl(aq) 2AlCl3(aq) + 3H2 (g). ln the lab, 0.15g H2 was collected. What is the % yield of the...
-
A 15.0-mL sample of an oxalic acid solution requires 25.2mL of 0.149 M NaOH for neutralization. Calculate the volume of a 0.122 M KMnO4 solution needed to react with a second 15.0-mL sample of the...
-
True or False: 1. Economists are not completely in agreement on what constitutes money for all purposes. 2. People use nontransaction accounts primarily because they generally pay higher interest...
-
Collegiate Corn Hole is a small business that Zach Morris developed while in college. He began building wooden corn hole game sets for friends, hand painted with college colors and logos. As demand...
-
Starbooks Corporation provides an online bookstore for electronic books. The following is a simplified list of accounts and amounts reported in its accounting records. The accounts have normal debit...
-
The Accounting Department at van Dyke Industries has produced a data set of the companys quarterly sales revenue for the past five years in an unstacked arrangement. These data are provided in the...
-
The partial trial balances of P Co. and S Co. at December 31, Year 5, were as follows: Additional Information ¢ The investment in the shares of S Co. (a 90% interest) was acquired January 2,...
-
Describe the advantages and disadvantages of the three temporal orientations - past, present, and future. How do these temporal perspectives differ across culture?
-
Peru Furniture City, Inc., sold $88,000 of furniture in May to customers who used their American Express Credit Cards. Such sales are subject to a 5% discount by American Express (a nonbank credit...
-
I am writing a recommendation report about office furnace. Can you show me how can I do three visuals? the visuals I have to do are about the following Desk Option 1: L-Shaped Desk An L-Shaped Desk...
-
For each of the following transactions for New Idea Corporation, prepare the adjusting journal entries required on July 31. (If no entry is required for a transaction/event, select "No Journal Entry...
-
1- How much force does it take to move a 137.3 kg box with an acceleration of 8.2 ms 2 ? 2- A projectile is launched from a height of 19.8 m, with an initial velocity of 17.9 in the horizontal...
-
How do modern operating systems manage concurrency in multi-core and multi-processor environments? Illustrate with examples of synchronization techniques and their impact on system performance .
-
XYZ Ltd owns a land-based drilling rig. It is in near perfect condition and unmodified. The company wishes to establish a fair value for the rig and identified two markets where near identical rigs...
-
Workers around jet aircraft typically wear protective devices over their ears. Assume that the sound level of a jet airplane engine, at a distance of 27 mm, is 130 dBdB, and that the average human...
-
Lanier Company began operations on January 1, 2014, and uses the FIFO method in costing its raw material inventory. Management is contemplating a change to the LIFO method and is interested in...
-
A supermarket chain is interested in exploring the relationship between the sales of its store-brand canned vegetables (y), the amount spent on promotion of the vegetables in local newspapers (x1)...
-
At STP, 0.280 L of a gas weighs 0.400 g. Calculate the molar mass of the gas.
-
The alcohol content of hard liquor is normally given in terms of the "proof," which is defined as twice the percentage by volume of ethanol (C2H5OH) present. Calculate the number of grams of alcohol...
-
How many carbon-carbon sigma bonds are present in each of the following molecules? (a) 2-butyne. (b) Anthracene (c) 2,3-dimethylpentane
-
When a honeybee flies through the air, it develops a charge of +17 pC. How many electrons did it lose in the process of acquiring this charge?
-
Falling raindrops frequently develop electric charges. Does this create noticeable forces between the droplets? Suppose two 1.8 mg drops each have a charge of +25 pC; these are typical values. The...
-
A housefly walking across a surface may develop a significant electric charge through a process similar to frictional charging. Suppose a fly picks up a charge of +52 pC. How many electrons does it...

Study smarter with the SolutionInn App


