2 NO2(g). If 0.30 mol of N2O4 is A.2. At certain temperature Kc= 4.50 for the...
Fantastic news! We've Found the answer you've been seeking!
Question:
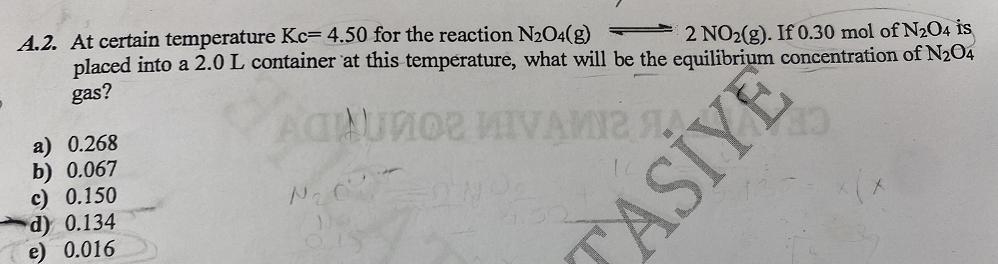
Transcribed Image Text:
2 NO2(g). If 0.30 mol of N2O4 is A.2. At certain temperature Kc= 4.50 for the reaction N₂O4(g) placed into a 2.0 L container at this temperature, what will be the equilibrium concentration of N₂04 gas? a) 0.268 b) 0.067 c) 0.150 d) 0.134 e) 0.016 720 ACAUNO MIVAMIZ A N₂C²=ON ASIYE 2 NO2(g). If 0.30 mol of N2O4 is A.2. At certain temperature Kc= 4.50 for the reaction N₂O4(g) placed into a 2.0 L container at this temperature, what will be the equilibrium concentration of N₂04 gas? a) 0.268 b) 0.067 c) 0.150 d) 0.134 e) 0.016 720 ACAUNO MIVAMIZ A N₂C²=ON ASIYE 2 NO2(g). If 0.30 mol of N2O4 is A.2. At certain temperature Kc= 4.50 for the reaction N₂O4(g) placed into a 2.0 L container at this temperature, what will be the equilibrium concentration of N₂04 gas? a) 0.268 b) 0.067 c) 0.150 d) 0.134 e) 0.016 720 ACAUNO MIVAMIZ A N₂C²=ON ASIYE
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below The image shows a chemistry problem related to chemical equilibrium The chemical reaction ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
A rigid container holds 4 kg of water at 20C. If 200 kJ of heat is added to this system, what will be the final temperature? How much work is done in this process?
-
Calculate the labour turnover rate according to replacement method from the following: No. of workers on the payroll: - At the beginning of the month: 500 - At the end of the month: 600 During the...
-
You are a new junior accountant at In Focus Corporation, maker of lenses for eyeglasses. Your company sells generic-quality lenses for a moderate price. Your boss, the controller, has given you the...
-
Golf Academy, Inc., provides private golf lessons. Its unadjusted trial balance at December 31, 2015, follows, along with information about selected accounts. Required: 1. Calculate the (preliminary)...
-
Laghari & Lagharis Personnel Department maintains a file of all employees of the company and each employees unique Employee ID. The file llemployees provides information on employees for Laghari &...
-
Poole Company began the 2013 accounting period with $36,000 cash, $80,000 inventory, $70,000 common stock, and $46,000 retained earnings. During the 2013 accounting period, Poole experienced the...
-
Suppose that a person has an average heart rate of 62.0 beats/min. How many beats does he or she have in 1.0 y? Incorrect not valid decimal or scientific notation. In 1.00 y?
-
What type of web server is sending packet/frame 280 of the capture? 2. What type of HTTP Status Code is the web server in packet/frame 314 sending? Google the error and provide a quote summarizing...
-
Church commenced business on 1st January 2021 with a Capital of 150,000 in cash. On the same date Church opened a bank account and deposited 30,000. During the month of January 2021 the following...
-
A 8,700-kg truck runs into the rear of a 1,200-kg car that was stationary. The truck and car are locked together after the collision and move with speed 3.5 m/s. Compute how much kinetic energy was...
-
1 Opening Balance Sheet The Wired Cup Assets Liabilities Current Assets: Current Liabilities: Cash $ 5,000 Bank Note $ 3,371 Accounts Receivable $ - Accounts Payable $ - Pre-Opening Expenses...
-
Let u = (2,1,1,2), a = (4,-4,2,-2). Find projau and u-projau.
-
Factor. 25v230v+9
-
Two manned satellites approach one another at a relative speed of 0.190 m/s, intending to dock. The first has a mass of 4.70 10 3 kg, and the second a mass of 7.90 10 3 kg. If the two satellites...
-
A machine costing $215,400 with a four-year life and an estimated $19,000 salvage value is installed in Luther Company's factory on January 1. The factory manager estimates the machine will produce...
-
The cost curve for the city water supply is C(Q) = 16 + 1/4 Q2, where Q is the amount of water supplied and C(Q) is the cost of providing Q acre-feet of water. (An acre-foot is the amount of water...
-
The concentration of Mg2+ in seawater is 0.052 M. At what pH will 99% of the Mg2+ be precipitated as the hydroxide salt? [Ksp for Mg(OH)2 = 8.9 10-12.]
-
Is the following statement true or false? Explain your answer. When determining the molar mass of a solute using boiling-point or freezing-point data, camphor would be the best solvent choice of all...
-
The freezing point of t-butanol is 25.50oC and Kf is 9.1oC kg/mol. Usually t-butanol absorbs water on exposure to air. If the freezing point of a 10.0-g sample of t-butanol is 24.59oC, how many grams...
-
The number on the jersey of each New York Giants football player is recorded, then the mean of those numbers is computed. Decide whether the statement makes sense (or is clearly true) or does not...
-
A professor calculates final grades using a weighted mean in which the final exam counts twice as much as the midterm. Decide whether the statement makes sense (or is clearly true) or does not make...
-
The following ages (years) of survey respondents: 22, 19, 21, 27, over 65, over 80. State whether the mean or median would give a better description of the average. Explain your reasoning.

Study smarter with the SolutionInn App


