5 liters of air with a humidity of 0.6, a temperature of 20 C and a...
Fantastic news! We've Found the answer you've been seeking!
Question:
5 liters of air with a humidity of 0.6, a temperature of 20 ° C and a pressure of 75 cm Hg it is pressed together to 2 liters at a temperature of 25 ° C.
Calculate all missing state parameters, the mass of dry air and the masses of water vapor and water in each state.
Bee what volume at 25 ° C is the considered air saturated with water vapor?
The atmosphere\'s air consists of approximately 78% by volume nitrogen gas, 21% by volume oxygen gas and almost 1% argon.
Air has a theoretical molar mass of 28.97 g / mol.
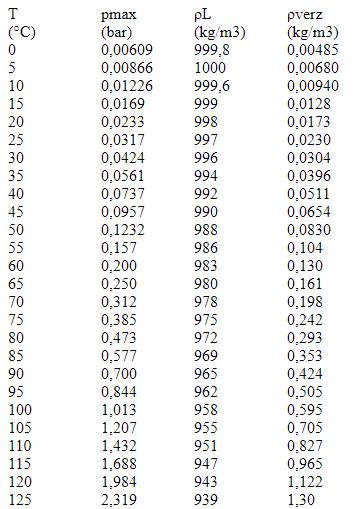
Table of Maximum pressure, density for water and saturated water vapor
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:




