5.0 g of aniline (C6H5NH2) is injected into an empty 6.5 L vessel at 140 C....
Fantastic news! We've Found the answer you've been seeking!
Question:
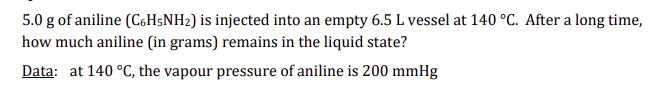
Transcribed Image Text:
5.0 g of aniline (C6H5NH2) is injected into an empty 6.5 L vessel at 140 °C. After a long time, how much aniline (in grams) remains in the liquid state? Data: at 140 °C, the vapour pressure of aniline is 200 mmHg 5.0 g of aniline (C6H5NH2) is injected into an empty 6.5 L vessel at 140 °C. After a long time, how much aniline (in grams) remains in the liquid state? Data: at 140 °C, the vapour pressure of aniline is 200 mmHg
Expert Answer:
Answer rating: 100% (QA)
1 Number of moles of aniline Number of moles 50 g 9313 gmol Numbe... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Let A, B be sets. Define: (a) the Cartesian product (A B) (b) the set of relations R between A and B (c) the identity relation A on the set A [3 marks] Suppose S, T are relations between A and B, and...
-
"internet radios" for streaming audio, and personal video recorders and players. Describe design and evaluation processes that could be used by a start-up company to improve the usability of such...
-
While you may not work for a large corporation, there is merit to the notion that the organization must speak with one voice and speak quickly in a crisis. What forces (tradition, practice, and...
-
Simpson, Locke, and Job each have a $27,000 Capital balance. Simpson is retiring from the business. The partners agree to revalue the assets at current market value. A real estate appraiser values...
-
Define the term "total retail experience." Then describe a recent retail situation in which your expectations were surpassed and state why.
-
Assume the same information as in question 4. Also assume that beginning work in process had \($6,000\) in conversion cost and that \($84,000\) in conversion is added during this period. What is the...
-
Rodgers Corporation produces and sells football equipment. On July 1, 2014, Rodgers Corporation issued $65,000,000 of 10-year, 12% bonds at a market (effective) interest rate of 10%, receiving cash...
-
Mercedes-Benz AG is a subsidiary of Daimler AG known for producing luxury vehicles and commercial vehicles. This company has strongly pushed for product placement in Hollywood movies. In 2015,...
-
Identify by letter the curve you would expect in the titration of a solution containing (a) Disodium maleate, Na 2 M, with standard acid. (b) Pyruvic acid, HP, with standard base. (c) Sodium...
-
The data related to JK company is given below: Income tax payable $61,250 $17,500 $24,400 $5,000 $4,320 $6,000 Allowance for doubtful accounts Accounts payable Interest receivable Unearned revenue...
-
Ravsten Company uses a job-order costing system. On January 1, the beginning of the current year, the companys inventory balances were as follows: Raw materials $ 19,000 Work in process $ 11,200...
-
Discuss how you would specifically prepare for trial. What questions do you anticipate being asked, and how would you answer them? Does your analysis uncover any investigative mistakes or problem...
-
1. What roles did you see emerge from your group project? What type of roles were they (social roles, task roles, dysfunctional roles)? What behaviors did the individuals exhibit that indicated they...
-
The distance from the Earth to Pluto is 4.67times 10^(9) miles. If a new flying machine can travel 1.92times 10^(5) miles per year, how many years would it take to reach Pluto? Write your answer in...
-
Why does Zal have to call the Simurgh to help Rudabeh when she is giving birth to ustem?
-
The correct increasing bond order of the following species 0, (A), 0 (B), CO (C), NO (D)will be (A) (A) < (B) < (D) < (C) (B) (A) < (B) < (C) < (D) (C) (D) < (C) < (B) < (A) (D) (B) < (A) < (D) < (C)
-
What are the key elements of a system investigation report?
-
A first-order heterogeneous irreversible reaction is taking place within a spherical catalyst pellet that is plated with platinum throughout the pellet. The reactant concentration halfway between the...
-
The irreversible first-order (wrt partial pressure of A) gas-phase reaction A B is carried out isothermally in a fluidized catalytic CSTR containing 50 kg of catalyst. Currently, 50% conversion is...
-
Example 7-1. What is the error in assuming the concentration of species B is constant and what limits can you put on the calculated value of k? (i.e., k = 0.24 ?) Example 7-1 The liquid-phase...
-
A Consider \[\begin{cases}\dot{x}_{1}=-x_{1}\left|x_{1} ight|+a x_{2}-\frac{2}{3}+\sin t & x_{1}(0)=\frac{2}{3} \\ \dot{x}_{2}=-x_{1}-\frac{1}{2} x_{2}-1, & x_{2}(0)=1\end{cases}\] where \(a\) is a...
-
The governing equation for an RLC circuit driven by the applied voltage \(v_{a}(t)\) is derived as \[L \frac{d i}{d t}+R i+\frac{1}{C} \int i d t=v_{a}(t)\] where \(L=4 \mathrm{H}, R=4 \Omega\), and...
-
The pendulum system in Figure 8.35 consists of a uniform thin rod of length \(l\) and a concentrated mass \(m\) at its tip. The friction at the pivot causes the system to be damped. When the angular...

Study smarter with the SolutionInn App


