50mL of 2% (W/W) solution of NaOH is neutralized completely with 0.50M H2SO4. Given a density...
Fantastic news! We've Found the answer you've been seeking!
Question:
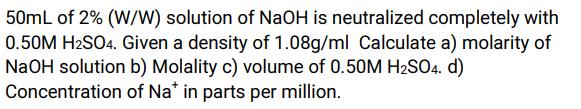
Transcribed Image Text:
50mL of 2% (W/W) solution of NaOH is neutralized completely with 0.50M H2SO4. Given a density of 1.08g/ml Calculate a) molarity of NaOH solution b) Molality c) volume of 0.50M H₂SO4. d) Concentration of Na* in parts per million. 50mL of 2% (W/W) solution of NaOH is neutralized completely with 0.50M H2SO4. Given a density of 1.08g/ml Calculate a) molarity of NaOH solution b) Molality c) volume of 0.50M H₂SO4. d) Concentration of Na* in parts per million.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the chemical shift in parts per million () for a proton that has resonance 128 Hz downfield from TMS on a spectrometer that operates at 60 MHz.
-
The hydrocarbon emissions at idling speed in parts per million (ppm) for automobiles of 1980 and 1990 are given for 20 randomly selected cars. (a) Construct a dot plot as in Figure 1.1. (b) Compute...
-
Atmospheric contaminants are often measured in parts per million (by volume). What would the partial pressure of refrigerant-134a be in atmospheric air at 100 kPa and 20oC to form a 100-ppm...
-
Patagonia founder and owner Yvon Chouinard argued that meaningful work depends on what one does, in terms of daily tasks, but also on the larger issues that a company stands for" He writes....
-
Determine the unknown quantity for each of the following independent situations using the appropriate interest tables: 1. Sue wants to have $10,000 saved when she begins college. If Sue enters...
-
In a model with an Allee effect, organisms reproduce poorly when the population is small. In one case, per capita production follows Our models of bacterial population growth neglect the fact that...
-
On January 1, 2020, Josie Inc. purchased for cash ten, \(\$ 1,000,4 \%\) bonds of Star Corp. at par. The bond interest is paid annually on January 1 of each year, and the bonds mature on January 1,...
-
Frandec Company manufactures, assembles, and rebuilds material handling equipment used in warehouses and distribution centers. One product, called a Liftmaster, is assembled from four components: a...
-
VICTORIA COMPANY CVP Income Statement For the Month Ended April 30, 2020 Total Per Unit Sales (10,000 units) $470,000 $47 Variable costs 220,900 22.09 Contribution margin 249,100 $24.91 Fixed...
-
(a) A horizontal steel I-beam of cross-sectional area 0.041m2 is rigidly connected to two vertical steel girders. If the beam was installed when the temperature was 30oC, what stress is developed in...
-
Describe the concept of "money as a weapon" and the associated authorities, factors, and decision methodology involved in enhancing force capability. Provide an example(s) or scenario(s) depicting...
-
Using EDGAR (Electronic Data Gathering, Analysis, and Retrieval system), find the annual report (10-K) for Six Flags Entertainment Corporation for the year ended December 31, 2019. Locate the...
-
Vigo Vacations has $300 million in total assets, $7 million in notes payable, and $44 million in long- term debt. What is the debt ratio? Do not round intermediate calculations. Round your answer to...
-
Five years ago, State X bought Railroad (RR), which was in bankruptcy and about to be liquidated. RR has always been the largest rail carrier in State X, presently carrying 70% of its rail freight....
-
S You are a relatively recent hire to Hartz & Company, a local manufacturer of plumbing supply products. You have been asked to prepare a condensed statement of cash flows for the months of November...
-
A 0.045 kg green marble collides with a stationary 0.070 kg yellow marble. Before the collision, the green marble was moving with a speed of 3.1 m/s, but after the collision, the green marble had a...
-
a) 2 year monthly pay annuity immediate whose payments grow geometrically at rate R per period. Her initial payment is $100.00 and the nominal annual interest rate is 9.20% compounded annually. The...
-
DC has unused FTC carryover from 2017 in the separate category for GC income as the result of income generated by a foreign branch. The income was foreign source general category income. In 2018 the...
-
The activity series in Section 4.4 shows that reaction (a) is spontaneous while reaction (b) is non spontaneous at 25C: (a) Fe(s) + 2H+ Fe2+(aq) + H2(g) (b) Cu(s) + 2H+ Cu2+(aq) + H2(g) Use the data...
-
Ethylene (C2H4), an important industrial organic chemical, can be prepared by heating hexane (C6H14) at 8008C: If the yield of ethylene production is 42.5 percent, what mass of hexane must be reacted...
-
A freshly isolated sample of 90Y was found to have an activity of 9.8 105 disintegrations per minute at 1:00 p.m. on December 3, 2003. At 2:15 p.m. on December 17, 2003, its activity was...
-
Puffin Industries acquired 90 percent of Sunset Coast Digitals stock on January 1, 2014, for \($3,150,000.\) At that time, Sunset Coasts stockholders equity totaled \($1,400,000,\) and the estimated...
-
Bottling Company acquired a 75 percent interest in Regional Bottling Partnership on January |, 2008. Assume the following information related to this acquisition: The excess of fair value over book...
-
Comparative consolidated balance sheets and the intervening income statement for Prime Casinos and its subsidiary Saratoga International Hotels are shown below: Additional information (in millions):...

Study smarter with the SolutionInn App


