6. The water-gas shift reaction (WGSR) is an important industrial reaction that is used in the...
Fantastic news! We've Found the answer you've been seeking!
Question:
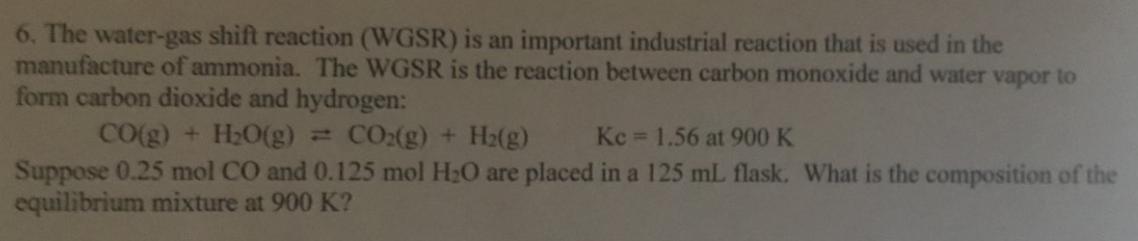
Transcribed Image Text:
6. The water-gas shift reaction (WGSR) is an important industrial reaction that is used in the manufacture of ammonia. The WGSR is the reaction between carbon monoxide and water vapor to form carbon dioxide and hydrogen: CO(g) + H20(g) = CO:(g) + H2(g) Kc 1.56 at 900 K Suppose 0.25 mol CO and 0.125 mol H20 are placed in a 125 mL flask. What is the composition of the equilibrium mixture at 900 K? 6. The water-gas shift reaction (WGSR) is an important industrial reaction that is used in the manufacture of ammonia. The WGSR is the reaction between carbon monoxide and water vapor to form carbon dioxide and hydrogen: CO(g) + H20(g) = CO:(g) + H2(g) Kc 1.56 at 900 K Suppose 0.25 mol CO and 0.125 mol H20 are placed in a 125 mL flask. What is the composition of the equilibrium mixture at 900 K?
Expert Answer:
Related Book For 

Thermodynamics An Interactive Approach
ISBN: 978-0130351173
1st edition
Authors: Subrata Bhattacharjee
Posted Date:
Students also viewed these accounting questions
-
The strength of paper used in the manufacture of cardboard boxes (y) is related to the percentage of hardwood concentration in the original pulp (x). Under controlled conditions, a pilot plant...
-
A machine makes rivets that are used in the manufacture of airplanes. To be acceptable, the length of a rivet must be between 0.9 centimeter and 1.1 centimeters. The mean length of a rivet is 1.0...
-
A press produces parts used in the manufacture of large-screen plasma televisions. If the press is correctly adjusted, it produces parts with a scrap rate of 5%. If it is not adjusted correctly, it...
-
In the Edgeworth production box diagram, what conditions must hold for an allocation to be on the production contract curve? Why is a competitive equilibrium on the contract curve?
-
A company manufacturing CDs is working on a new technology. A random sample of 703 Internet users were asked: As you may know, some CDs are being manufactured so that you can only make one copy of...
-
On January 2, 2012, Kowalchuk Corporation, a small company that follows ASPE, issued $1.5 million of 10% bonds at 97 due on December 31, 2021. Legal and other costs of $110,000 were incurred in...
-
Continuing Question 5, turn off the lag filter and use a notch filter to improve the performance. Simultaneously adjust Notch Freq and KVP to maximize KVP without inducing peaking in the closed-loop...
-
The Buffalo Snow Shoe Company is considering manufacturing radial snow shoes, which are more durable and offer better traction. Buffalo estimates that the investment in manufacturing equipment will...
-
Explain how convergence differs from harmonization. In your opinion, what are the two most important arguments in favor of international convergence for US public companies? In your opinion, what are...
-
The accounts in the ledger of Time Is Money Delivery Service contain the following balances on July 31, 2015. Accounts Receivable ......... $10,642 Prepaid Insurance ............ $ 1,968 Accounts...
-
Use a formula, or a table and/or a graph to decide whether the limit exists. If it exists, find its value. As discussed in the class, you can use the formula = In (a). lim x10 01 02 12212 (-x-1-2x-1)...
-
What organizational capabilities are required to support the launch of "Command Products"?
-
What type of team is made up of people pulled from various departments, but they still have to do their departmental job in addition to being on the innovation team?
-
Discuss the Triple A Supply Chain article outlining the definition of each strategy and provide methods in what is the companies can meet those objectives?
-
What hurdles is InBev going to face to complete its acquisition of AB? What can InBev do to overcome these hurdles? 4) What options does AB have if it wants to continue as an independent player? 5)...
-
Explain how Release of HIPAA information manager will need to adjust and adapt to overcome these challenges and avoid future risks?
-
A crate required 900 newtons (N) of force to lift. The crate was lifted by a forklift with an engine that provided 225 N of force. What is the mechanical advantage of the forklift?
-
Three successive resonance frequencies in an organ pipe are 1310, 1834, and 2358 Hz. (a) Is the pipe closed at one end or open at both ends? (b) What is the fundamental frequency? (c) What is the...
-
A symmetric converging diverging duct with an area ratio of 2 is placed in wind tunnel where it encounters a 700 m/s flow of air at 50 kPa and 300 K. Determine the bypass ratio (diverted flow /...
-
Derive an expression for estimating the pressure (p) at which graphite and diamond exist in equilibrium at 300 K and 100 kPa in terms of the specific Gibbs function.
-
A refrigerant is compressed by an adiabatic compressor operating at steady state to raise the pressure from 200 kPa to 750 kPa. The following data are supplied for the inlet and exit ports. Inlet: v...
-
Explain why budgets are important to all organizations. Expand this discussion by illustrating how different types of budgets are used.
-
Compare and contrast licensure and credentialing.
-
Describe the steps in attaining state licensure for nurses.

Study smarter with the SolutionInn App



