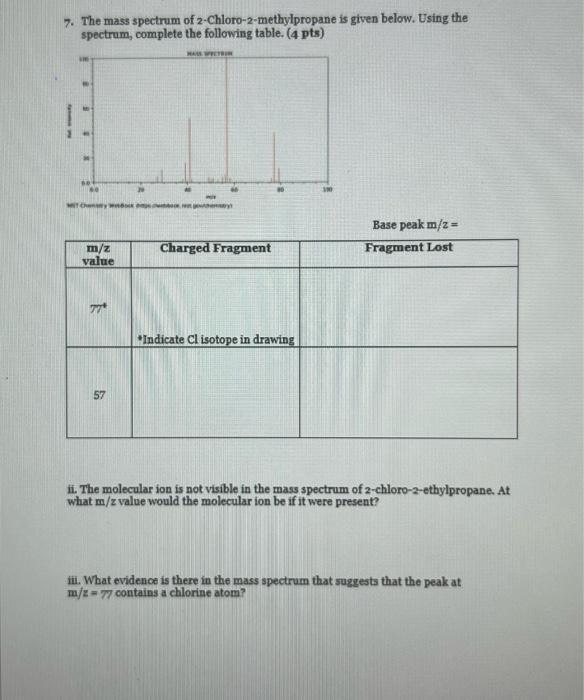
7. The mass spectrum of 2-Chloro-2-methylpropane is given below. Using the spectrum, complete the following table....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
7. The mass spectrum of 2-Chloro-2-methylpropane is given below. Using the spectrum, complete the following table. (4 pts) MASS SPECTRIN 60 Che m/z value 1 Widok o 57 goty Charged Fragment *Indicate Cl isotope in drawing Base peak m/z = Fragment Lost ii. The molecular ion is not visible in the mass spectrum of 2-chloro-2-ethylpropane. At what m/z value would the molecular ion be if it were present? iii. What evidence is there in the mass spectrum that suggests that the peak at m/z-77 contains a chlorine atom? 7. The mass spectrum of 2-Chloro-2-methylpropane is given below. Using the spectrum, complete the following table. (4 pts) MASS SPECTRIN 60 Che m/z value 1 Widok o 57 goty Charged Fragment *Indicate Cl isotope in drawing Base peak m/z = Fragment Lost ii. The molecular ion is not visible in the mass spectrum of 2-chloro-2-ethylpropane. At what m/z value would the molecular ion be if it were present? iii. What evidence is there in the mass spectrum that suggests that the peak at m/z-77 contains a chlorine atom?
Expert Answer:
Answer rating: 100% (QA)
solution 598 chloro2methyl propane Cl Answer i m2 77 57 ctfCCH 35 1277 598 35C m277 arc C03 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these physics questions
-
In contrast to 2-methylbutane and neopentane, the mass spectrum of 3-methylpentane (not given) has a peak of very low relative abundance at - 15. It has a peak of very high relative abundance at -...
-
The molecular ion region in the mass spectrum of a large molecule, such as a protein, consists of a cluster of peaks differing by 1 Da. This pattern occurs because a molecule with many atoms has a...
-
The mass spectrum of unknown compound A shows a molecular ion at m/z 116 and prominent peaks at m/z 87 and m/z 101. Its UV spectrum shows no maximum above 200 nm. The IR and NMR spectra of A follow....
-
Provide a detailed description of the JDBC three-tier model and its function.
-
Meek, Inc., remodeled its offices this year. Renee, the executive vice president, bought the desk, couch, and lamp set that had been in her office for $200. Identify the tax issue(s) posed by the...
-
Is it possible for a monopolistically competitive firm to continue to earn an economic profit as new firms enter the market?
-
Peppertree Copy Center had accounts receivable of \(\$ 20,000\) at the beginning of the year and \(\$ 50,000\) at year-end. Revenue for the year totaled \(\$ 110,000\). How much cash did Peppertree...
-
Professor Patricia (Patty) Pa^te is retired from the Palm Springs Culinary Arts Academy (PSCAA). She is a single taxpayer and is 68 years old. Patty lives at 98 Colander Street, Henderson, NV 89052....
-
Not-for-sale" is a California-based organization whose aim is to eliminate modern slavery while "Free2Work" (its sister group organization) collects the data and makes assessment based on three...
-
Complete Tsate's Form 1040-SR, Schedules A, B and D, Form 8949, Form 6252 and Qualified Dividends and Capital Gain Tax Worksheet. Tsate Kongia (birthdate 02/14/1954) is an unmarried high school...
-
Determine the solubility of the ions that is calculated from the Ksp for Mg2 CO3. 2,53 OS 2 O 4.5 3 2.5 2 S3
-
Based on the passage titled "The Problem of Cocolisap " used in Discovery, what is the type of noun according to characteristic? How do you know if it's ominous or special? Write your answer in the...
-
Suppose F = grad(6x - e). In each case, find the value of C (a) C is any curve in the xy-plane from (0, 2) to (5, 0). 7.07 =[ .dr
-
Municipality A has a population of approximately 90 000 with more or less 18000 households. Approximately 69% of the population resides in the local township area. Just over half of the Municipality...
-
Currently the federal reserve discount rate is within the range 1.00% to 1.25%. At which of these two discount rate bounds the price of a 100,000 face value 90 days money market securities is higher?...
-
Evaluate the integral. 3xe5x dx
-
Put yourself in the shoes of a Tesla shareholder. On June 21, 2016, Tesla announced that it made an all-equity offer to acquire SolarCity. Because this transaction will require a substantial amount...
-
Assume that a trial balance is prepared with an account balance of $21,360 listed as $21,630 and an account balance of $1,500 listed as $15,000. Identify the transposition and the slide.
-
(a) The isoelectric point (pI) of phenylalanine is pH 5.5. Draw the structure of the major form of phenylalanine at pH values of 1, 5.5, and 11. (b) The isoelectric point of histidine is pH 7.6. Draw...
-
(A true story.) Chemistry lab students added an excess of ethylmagnesium bromide to methyl furoate, expecting the Grignard reagent to add twice and form the tertiary alcohol. After water workup, they...
-
The spectrum of trans-hex-2-enoic acid follows. (a) Assign peaks to show which protons give rise to which peaks in the spectrum. (b) Draw a tree to show the complex splitting of the vinyl proton...
-
Find the area under the standard normal curve to the right of a. z = 0.47 b. z = 2.91 c. z = 2.04 d. z = 1.09
-
Find the area under the standard normal curve that lies outside the interval between a. z = 0.38 and z = 1.02 b. z = 1.42 and z = 1.78 c. z = 0.01 and z = 2.67 d. z = 2.45 and z = 0.34
-
Find the area under the standard normal curve that lies outside the interval between a. z = 1.11 and z = 3.21 b. z = 1.93 and z = 0.59 c. z = 0.46 and z = 1.75 d. z = 2.73 and z = 1.39

Study smarter with the SolutionInn App


