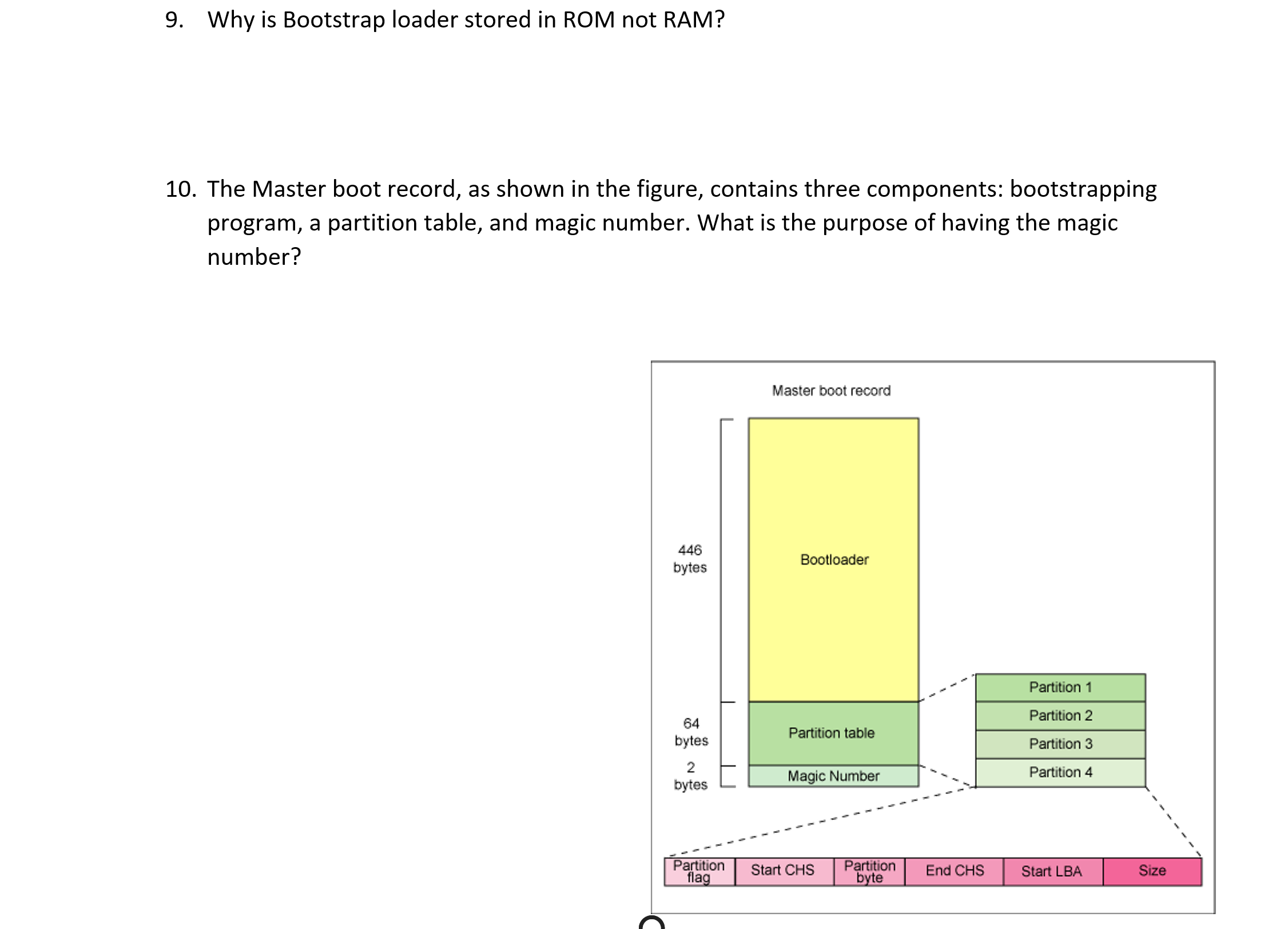
9. Why is Bootstrap loader stored in ROM not RAM? 10. The Master boot record, as...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
9. Why is Bootstrap loader stored in ROM not RAM? 10. The Master boot record, as shown in the figure, contains three components: bootstrapping program, a partition table, and magic number. What is the purpose of having the magic number? 446 bytes 64 bytes 2 bytes Partition flag Master boot record Bootloader Partition table Magic Number Start CHS Partition byte End CHS Partition 1 Partition 2 Partition 3 Partition 4 Start LBA Size 9. Why is Bootstrap loader stored in ROM not RAM? 10. The Master boot record, as shown in the figure, contains three components: bootstrapping program, a partition table, and magic number. What is the purpose of having the magic number? 446 bytes 64 bytes 2 bytes Partition flag Master boot record Bootloader Partition table Magic Number Start CHS Partition byte End CHS Partition 1 Partition 2 Partition 3 Partition 4 Start LBA Size
Expert Answer:
Answer rating: 100% (QA)
The question asks about two different things related to the booting process of a computer 1 Why is t... View the full answer

Related Book For 

Systems analysis and design
ISBN: 978-0136089162
8th Edition
Authors: kenneth e. kendall, julie e. kendall
Posted Date:
Students also viewed these programming questions
-
ualitative Assessment of the Risks of Individual Assets. To answer this question, you should think about what kinds of risk each stock faces (based on the firm descriptions in the case). There are no...
-
Explain the purpose of the Master Boot Record (MBR) as itrelates to providing an OS with information about existingpartitions. Emphasize that because the MBR is limited to devicesthat are less than...
-
My question is how should this look. Below this is my chart but not sure if I am corrrectly? The purpose of this assignment is to practice on the creation of a Relational Data Model from an Entity...
-
Verify that A and AÌ = p -1 AP have the same spectrum. 19 4 2 12 1 P.
-
The standard potentials of proteins are not commonly measured by the methods described in this chapter because proteins often lose their native structure and function when they react on the surfaces...
-
Think about the most recent technological developments. What are the likely implications of these developments for employers during the next 10 years? For employees?
-
The performance characteristics of a certain centrifugal pump are determined from an experimental setup similar to that shown in Fig. 12.10. When the flowrate of a liquid \((S G=0.9)\) through the...
-
This problem continues the Draper Consulting, Inc., situation from P21-35 of Chapter 21. Assume Draper Consulting began January with $29,000 cash. Management forecasts that collections from credit...
-
Please help describe one intervention that used a health behavior theory or model, that was effective in modifying the health behavior of high risk sexual behavior?
-
The following rates were observed for a first-order, irreversible reaction, carried out on a spherical catalyst: dp = 0.625 cm robs=0.09 mol/gcat.hr dp = 0.10 cm Tobs = 0.275 mol/gcat.hr Strong...
-
Which of the following are debt instruments that companies offer as investments? a. bonds b. stocks c. bank loans d. unpaid accounts
-
Following the discussion prompt this week, I looked in the National Institute of Justice Database for an article. Using the filter to find topics involving probability and choosing amongst scholarly...
-
Design a function utilizing a learning algorithm with the error-correction learning rule. The function shall be robust, so it should not depend on the length of the input, the size of a learning set,...
-
1. How would test your set-up if it is in equilibrium? Why?2. In your opinion, which method of determining the resultant is more efficient and practical or convenient touse? Defend your answer.3....
-
1.)Do you think that the Production Possibilities Frontier model is useful or not? Give some examples and explain your answer. 2.) We have two producers, Gwen and Blake, Gwen can produce 60 pizzas...
-
Assume that Pam's utility function is given by u = 40I 0.001I2, where I is income. Pam has the choice of legal work that pays IL = $10,000 per year or illegal work that pays II = $20,000 per year....
-
Write a net ionic equation to show that nitric acid, HNO3, behaves as an acid in water. Submit Answer + HO(1) Retry Entire Group + 5 more group attempts remaining
-
Keating & Partners is a law firm specializing in labour relations and employee-related work. It employs 25 professionals (5 partners and 20 managers) who work directly with its clients. The average...
-
Think about how you would organize a data warehouse for Marathon Vitamin Shops. What other databases would you like to merge into the data warehouse? What sort of patterns should Bill Berry be...
-
Within your group, assign some of the roles that people take on in agile development. Make sure that one person is an onsite customer and at least two people are programmers. Assign other roles, as...
-
The five-year-old TowerWood hotel chain needs help designing its Web site. The company maintains properties in all the large U.S. tourist communities such as Orlando, Florida (near Disney World);...
-
With reference to Exercise 11.65, (a) find a \(99 \%\) confidence interval for the mean battery backup at \(x=1.25\); (b) find \(95 \%\) limits of prediction for the battery backup provided by a...
-
A chemical engineer found that by adding different amounts of an additive to gasoline, she could reduce the amount of nitrous oxides (NOx) coming from an automobile engine. A specified amount was...
-
To determine how well existing chemical analyses can detect lead in test specimens in water, a civil engineer submits specimens spiked with known concentrations of lead to a laboratory. The chemists...

Study smarter with the SolutionInn App


