A 0.0450 M solution of benzoic acid has a pH of 2.78. Calculate pK, for this...
Fantastic news! We've Found the answer you've been seeking!
Question:
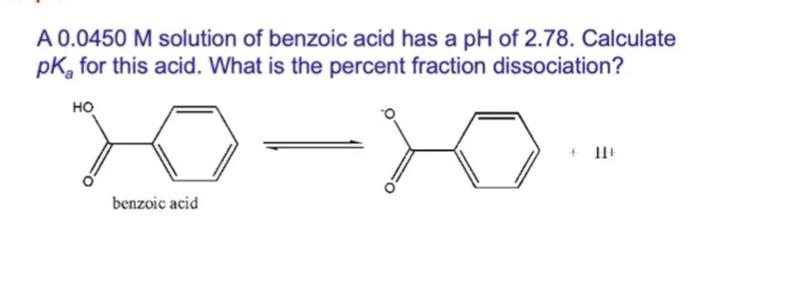
Transcribed Image Text:
A 0.0450 M solution of benzoic acid has a pH of 2.78. Calculate pK, for this acid. What is the percent fraction dissociation? но benzoic acid A 0.0450 M solution of benzoic acid has a pH of 2.78. Calculate pK, for this acid. What is the percent fraction dissociation? но benzoic acid
Expert Answer:
Answer rating: 100% (QA)
Solutiona chemicel Remetion Ho Conentiatiin ok Berzuic Avidu is D04 50M ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A 0.045 0 M solution of benzoic acid has a pH of 2.78. Calculate pKa for this acid.
-
A 0.0450 M solution of benzoic acid has a pH of 2.78. Calculate pK, for this acid. What is the percent fraction dissociation? benzoic acid
-
A 0.0450 M solution of benzoic acid has a pH of 2.78. Calculate pK, for this acid. What is the percent fraction dissociation? benzoic acid
-
Mary Sue owns 600 shares of QRS Moving Company. QRS pays a quarterly dividend of $0.50 per share. What is the total annual dividend that Mary Sue will receive?
-
A computer information systems professor is interested in studying the amount of time it takes students enrolled in the Introduction to Computers course to write a program in V B. NET. The professor...
-
A dogs bark has a sound intensity level of 40 dB. (a) If two of the same dogs were barking, the intensity level is (1) less than 40 dB, (2) between 40 dB and 80 dB, (3) 80 dB. (b) What would be the...
-
Consider a perpetual American put option (with \(T=\infty\) ). For small stock prices it will be advantageous to exercise the put. Let \(G\) be the largest such stock price. The time-independent...
-
On January 1, 2018, Allied Industries leased a high-performance conveyer to Karrier Company for a four-year period ending December 31, 2021, at which time possession of the leased asset will revert...
-
Suppose that a bushel of wool could be traded for a barrel of wine, one-for-one, on the international market. Draw a diagram illustrating the original situation, and this new situation, with wool on...
-
Write a paper about knowledge management strategy formulation this project provides an opportunity for you to identify opportunities for knowledge management at a real-world business and to suggest...
-
1 A CPA takes on a new individual tax client. The client drops off the information on February 10, 20xx. The CPA extends the return and notifies the client that it was extended. The client calls and...
-
Although the projects in this book are not necessarily aligned with educational goals, each has the potential to benefit from the participation of a subject matter expert. Try to find an expert in an...
-
Now is the time to begin evaluating your project, measuring it against marketing goals. Ask yourself the following questions. How is your design supporting or extending the brand experience? How is...
-
Look at any gamified system and identify where it exists on the gamified system spectrum. In what ways does the structure of the system and the messaging about it tell you if it is more or less like...
-
Research and play a gamified system and identify the characteristics of a gamified system. What do you think the goals are of the project members? In what ways is the system using game concepts? ...
-
Begin your project by finding out as much as you can about your audience and the games, gamified systems and technologies currently available to help them complete the tasks you are trying to...
-
When providing answers: - Please provide four-digit decimal numbers if your answer is in percentage. ex) If your answer is 12.36%, please provide 0.1236 If your answer is in dollars, please provide...
-
Grace is training to be an airplane pilot and must complete five days of flying training in October with at least one day of rest between trainings. How many ways can Grace schedule her flying...
-
Calculate E, G , and K for the following reactions. (a) 4Co3+ + 2H2O 4Co2+ O2(g) + 4H+ (b) Ag(S2O3)32- + Fe(CN)4-6 Ag(s) + 2S2O2- 3 + Fe(CN)3-6
-
Suppose that the Ag | AgCl outer electrode in Figure 14-11 is filled with 0.1 M NaCl instead of saturated KCl. Suppose that the electrode is calibrated in a dilute buffer containing 0.1 M KCl at pH...
-
(a) Explain the purpose of the collision cell in Figure 20-30. (b) Explain the purpose of a dynamic reaction cell, which can replace the collision cell. (c) In geologic strontium isotopic analysis,...
-
Personalized medicine may be used a. to characterize types of tumors. b. to predict the outcome of certain types of cancers. c. to determine the proper dosage of drugs. d. in all of the above.
-
Tumor-suppressor genes promote cancer when a. they are overexpressed. b. they are expressed in the wrong cell type. c. their function is inactivated. d. they are expressed at the wrong stage of...
-
Which of the following is a type of genetic change that could produce an oncogene? a. Missense mutation b. Gene amplification c. Chromosomal translocation d. All of the above can produce an oncogene.

Study smarter with the SolutionInn App


