A 0.06 g of the NaCl sample (FM: 58.44) was treated with 15.00 mL of standard...
Fantastic news! We've Found the answer you've been seeking!
Question:
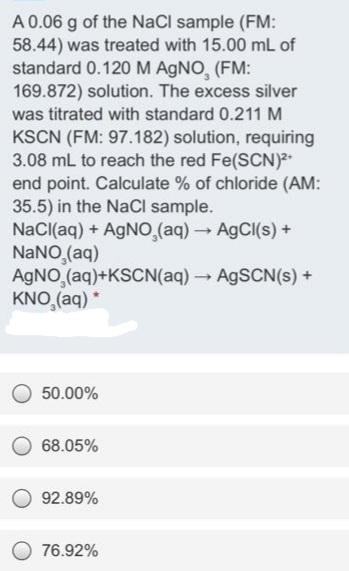
Transcribed Image Text:
A 0.06 g of the NaCl sample (FM: 58.44) was treated with 15.00 mL of standard 0.120 M AGNO, (FM: 169.872) solution. The excess silver was titrated with standard 0.211 M KSCN (FM: 97.182) solution, requiring 3.08 mL to reach the red Fe(SCN) end point. Calculate % of chloride (AM: 35.5) in the NaCl sample. NaCl(aq) + AGNO (aq)A9CI(s)+ NANO (aq) AGNO,(aq)+KSCN(aq) AGSCN(s) + KNO,(aq) * 50.00% 68.05% 92.89% 76.92% A 0.06 g of the NaCl sample (FM: 58.44) was treated with 15.00 mL of standard 0.120 M AGNO, (FM: 169.872) solution. The excess silver was titrated with standard 0.211 M KSCN (FM: 97.182) solution, requiring 3.08 mL to reach the red Fe(SCN) end point. Calculate % of chloride (AM: 35.5) in the NaCl sample. NaCl(aq) + AGNO (aq)A9CI(s)+ NANO (aq) AGNO,(aq)+KSCN(aq) AGSCN(s) + KNO,(aq) * 50.00% 68.05% 92.89% 76.92%
Expert Answer:
Answer rating: 100% (QA)
Ans 6805 Explanation From the given reaction AgNO 3 KSCN AgSCN KNO 3 we can see 1 AgNO 3 requires 1 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A solution of scandium chloride was treated with silver nitrate. The chlorine in the scandium compound was converted to silver chloride, AgCl. A 58.9-mg sample of scandium chloride gave 167.4 mg of...
-
A 20.0-mL sample of 0.200 M HBr solution is titrated with 0.200MNaOH solution. Calculate the pH of the solution after the following volumes of base have been added: (a) 15.0 mL, (b) 19.9 mL, (c) 20.0...
-
A 50.0-mL sample containing Ni2+ was treated with 25.0 mL of 0.050 0 M EDTA to complex all the Ni2+ and leave excess EDTA in solution. The excess EDTA was then back titrated, requiring 5.00 mL of...
-
Reference frame S is moving along the x axis at 0.6c relative to frame S. A particle that is originally at x = 10 m at t 1 = 0 is suddenly accelerated and then moves at a constant speed of c/3 in...
-
At the beginning of year 1, Ed and Fran each contributed $1,000 cash to EF Partnership as equal partners. The partnership immediately borrowed $98,000 on a nonrecourse basis and used the contributed...
-
As Figure 15-1 illustrates, the United States leads all OECD countries in health care expenditures, spending almost double the average OECD countrys share of GDP on health care. But American health...
-
An engineer prepares a report to evaluate a project using PW and IRR. Just before submitting the report, he spills coffee on it, making the first digit of the 2-digit IRR unreadable. The second digit...
-
The following direct materials and direct labor data pertain to the operations of Laurel Company for the month of August. Instructions (a) Compute the total, price, and quantity variances for...
-
Briefly describe the two Oracle Administrative Tools and talk about some of their administration usage: -SQL*Plus - Oracle Enterprise Manager Database Express (EM Express) Question2: Explain briefly...
-
Evie Excellent is trying to create financial statements for her start-up company, Stinky Soaps. Evie began the business on Jan 1, 2023 with a $1,000 investment from her parents. The year 2023 is...
-
Prepare an income statement for the year ending December 31, 2013, using the fixed cost data provided to improve the accuracy of the percentage of sales method. Prepare a balance sheet as of December...
-
You are one of Parlys lawyers prior to a planned meeting with isaiah and Tina , parly meets with you to advise icon on its strategy for forcing sushi me to comply with icons demands Which disclosure...
-
How can the topic of "elasticity" be related to agriculture. Please Cite references for study purposes.?
-
ASSESSMENT 2 - PROJECT STUDENT INFORMATION This information is to be handed to each student to outline the assessment requirements PART - A For this assessment, you are required to administer...
-
The circular arc shown below carries a charge per unit length = cos(6), where 0 is measured from the x-axis. What is the electric field at the origin? (Use the following as necessary: 10, 0, 0, r.) ...
-
To drive a typical car at 43 mph on a level road for one hour requires about 3.5 10^7 J of energy. Suppose we tried to store this much energy in a spinning, solid, uniform, cylindrical flywheel. A...
-
what are the benefits, risks and trade-offs to design a customer experience strategy which heavily relies on automation like machine learning, AI and BOTS to manage and serve customers and segment
-
Suppose the government bond described in problem 1 above is held for five years and then the savings institution acquiring the bond decides to sell it at a price of $940. Can you figure out the...
-
Predict the main product when HBr adds to H-C
-
Each of the following statements concerns a 0.10 M solution of a weak organic base, B. Briefly describe why each statement is either true or false. a. [B] is approximately equal to 0.10 M. b. [B] is...
-
A solution is prepared from 0.150 mol of formic acid and enough water to make 0.425 L of solution. a. Determine the concentrations of H3O+ and HCOO in this solution. b. Determine the H3O+...
-
What encoding scheme is extensive to represent all the characters of all the languages in the world?
-
What is hardware?
-
What is pseudocode?

Study smarter with the SolutionInn App


