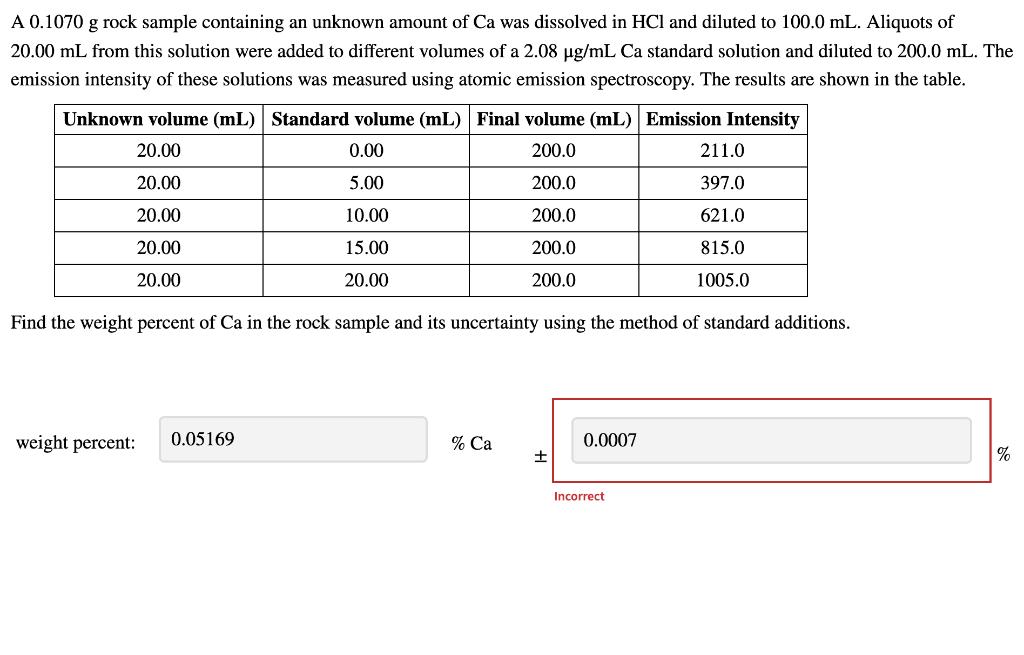
A 0.1070 g rock sample containing an unknown amount of Ca was dissolved in HCl and...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A 0.1070 g rock sample containing an unknown amount of Ca was dissolved in HCl and diluted to 100.0 mL. Aliquots of 20.00 mL from this solution were added to different volumes of a 2.08 ug/mL Ca standard solution and diluted to 200.0 mL. The emission intensity of these solutions was measured using atomic emission spectroscopy. The results are shown in the table. Unknown volume (mL) Standard volume (mL) Final volume (mL) Emission Intensity 20.00 0.00 200.0 211.0 20.00 5.00 200.0 397.0 20.00 10.00 200.0 621.0 20.00 15.00 200.0 815.0 20.00 20.00 200.0 1005.0 Find the weight percent of Ca in the rock sample and its uncertainty using the method of standard additions. weight percent: 0.05169 % Ca 0.0007 % Incorrect A 0.1070 g rock sample containing an unknown amount of Ca was dissolved in HCl and diluted to 100.0 mL. Aliquots of 20.00 mL from this solution were added to different volumes of a 2.08 ug/mL Ca standard solution and diluted to 200.0 mL. The emission intensity of these solutions was measured using atomic emission spectroscopy. The results are shown in the table. Unknown volume (mL) Standard volume (mL) Final volume (mL) Emission Intensity 20.00 0.00 200.0 211.0 20.00 5.00 200.0 397.0 20.00 10.00 200.0 621.0 20.00 15.00 200.0 815.0 20.00 20.00 200.0 1005.0 Find the weight percent of Ca in the rock sample and its uncertainty using the method of standard additions. weight percent: 0.05169 % Ca 0.0007 % Incorrect
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
An unknown amount of a radioactive substance is being studied. After two days, the mass is 15.231 grams. After eight days, the mass is 9.086 grams. How much was there initially? What is the half-life...
-
A sample of rock containing magnesite, MgCO3, was dissolved in hydrochloric acid, and the carbon dioxide gas that evolved was collected. If a 0.1504-g sample of the rock gave 37.71 mL of dry carbon...
-
A 0.0182-g sample of an unknown substance was dissolved in 2.135 g of benzene. The molality of this solution, determined by freezing-point depression, was 0.0698 m. What is the molecular mass of the...
-
Delivery Service purchased a commercial umbrella policy with a $10 million liability limit and a $100,000 self-insured retention. The umbrella insurer required Delivery Service to carry a $1 million...
-
At the beginning of the current year, Niles Corporation issued 1,600 shares of its own $ 2 par value common stock for $ 19 per share. Prepare the journal entry required to record the issuance of the...
-
Rain is falling on a poorly designed 150-m2 flat roof of a contemporary home. The design flaw requires the water level on the roof to reach approximately 5 cm above the plane of the roof before the...
-
Oxygen moves from the alveoli in the lungs into the bloodstream through the process of (a) active transport. (b) endocytosis. (c) diffusion. (d) exocytosis.
-
Deer Valley Kitchen, a fast-food restaurant company, operates a chain of restaurants across the nation. Each restaurant employs eight people; one is a manager paid a salary plus a bonus equal to 3...
-
1. What is the price of a $100 face value , 4year, zero -coupon bond if the market rate of interest of such risk free -zero coupon bonds is 5.95% 2. Consider a ten year bond with a face value of...
-
Your client mr. Smith has requested a reservation at eleven madison park this evening for a party of 2 at 6pm. Unfortunately, the restaurant is fully committed. As an alternative, which restaurant...
-
A firm aims to achieve a return on investment (ROI) of 25%. Compile a pro forma statement of financial performance and statement of financial position based on the budgeted figures below: The...
-
Beam \(A B C\) is supported by a three-bar truss at point \(C\) and at \(A\) by an elastomeric pad that is equivalent to a roller. (a) Compute the vertical deflection of point \(B\) in Figure P8.39...
-
Google, the immensely popular Web search engine, has been touted as the closest thing the Web has to an ultimate answer machine. Although this is debatable, of course, it is far more difficult to...
-
Discuss the relationship between GDP and the size of a nations services sector. Identify key causeand-effect relationships.
-
For the trusses in Figure P9. 29, compute the reactions and bar forces produced by the applied loads. Given: \(A E=\) constant, \(A=1000 \mathrm{~mm}^{2}\), and \(E=200 \mathrm{GPa}\). E 100 KN 60 KN...
-
The uses of pronouns in a statement are important for what major reason? a. Introduction b. Rude and impolite c. They are objective. d. They are personal.
-
Read this article (https://www.bankofcanada.ca/2020/12/understanding-quantitative-easing/) about understanding quantitative easing and answer the following. a. How is this approach different from...
-
The test statistic in the NeymanPearson Lemma and the likelihood ratio test statistic K are intimately related. Consider testing H 0 : = 0 versus H a : = a , and let * denote the test statistic...
-
Let a = (2, 2, 0), b = (1, -1, 1), and c = (-2, 2, 1). Find each of the following: a. a c b. (a - c) b c. a / ||a|| d. (b - c) a e. b c / ||b|| ||c|| f. a a - ||a||2
-
In statistics we defined the mean x and the variance s2 of sequence of number x1, x2, ... xn by Find x and s2 for the sequence of numbers 2, 5, 7, 8, 9, 10, 14 12. /t
-
Consider a country with a population of 10 million in 1985, a growth rate of 1.2% per year, and immigration from other countries of 60000 per year. Use the differential equation of problem 38 to...
-
A running mountain lion can make a leap 10.0 m long, reaching a maximum height of 3.0 m. a. What is the speed of the mountain lion just as it leaves the ground? b. At what angle does it leave the...
-
In punting a football, the kicker tries to maximize both the distance of the kick and its hang timethe time that the ball is in the air. A kicker gets off a great punt with a hang time of 5.0 s that...
-
Emily throws a soccer ball out of her dorm window to Allison, who is waiting below to catch it. If Emily throws the ball at an angle of 30 below horizontal with a speed of 12 m/s, how far from the...

Study smarter with the SolutionInn App


