A 0.1278 g sample of primary standard sodium oxalate required exactly 33.31 ml of the permanganate...
Fantastic news! We've Found the answer you've been seeking!
Question:
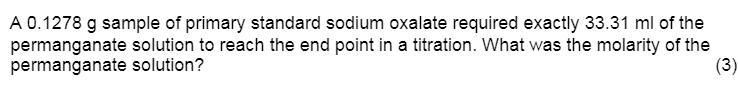
Transcribed Image Text:
A 0.1278 g sample of primary standard sodium oxalate required exactly 33.31 ml of the permanganate solution to reach the end point in a titration. What was the molarity of the permanganate solution? (3) A 0.1278 g sample of primary standard sodium oxalate required exactly 33.31 ml of the permanganate solution to reach the end point in a titration. What was the molarity of the permanganate solution? (3)
Expert Answer:
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Posted Date:
Students also viewed these chemistry questions
-
A 25.0-mL sample of sodium sulfate solution was analyzed by adding an excess of barium chloride solution to produce barium sulfate crystals, which were filtered from the solution. Na2SO4(aq) +...
-
A 1.00-L aqueous solution contained 5.80 g of sodium hydroxide, NaOH. What was the pH of the solution at 25C?
-
Exactly 75.00 mL of a 0.3132 M solution of Na2SO3 were treated with 150.0 mL of 0.4025 M HClO4 and boiled to remove the SO2 formed. (a) What was the mass in grams of SO2 that was evolved? (b) What...
-
Convex Mechanical Supplies produces a product with the following costs as of July 1, 2012: Material ............................... $ 6 Labor .................................. 4 Overhead...
-
In a balanced three-phase wye-wye system, the source is 20 + j1. The source has an abc-phase sequence and V an = 120 <0 o V rms. If the load voltage is V AN = 111.49
-
Match each term in Column A with its related definition in Column B. Column A 1. __________ Maquiladora 2. __________ Import 3. __________ Joint venture 4. __________ Export 5. __________ MNC Column...
-
During 2008, the world entered a global recession that resulted in many business failures. Today, companies continue to struggle with a lower degree of consumer confidence and lower levels of...
-
Assume Magnificent Modems, Inc. (MMI) is a division of Gilmore Business Products (GBP). GBP uses ROI as the primary measure of managerial performance. GBP has a desired return on investment (ROI) of...
-
4. Consider polynomial functions in the form f(x) = ax" + x. Use at least three examples to investigate the relationship between a, n, and the constant finite differences. Explain the steps you took...
-
As noted in Problem 8-24B, Watonga Swimsuit makes swimsuits. In 2018, Watonga produced its most popular swimsuit, the Sarong, for a standard labor price of $33.60 per hour. The standard amount of...
-
. X & Y are desirous to purchase a business andyou are asked to advise the average amount of working capital which will be required in the first year's working from the following estimates.Also add...
-
You have to decide whether to invest $100 in a friend's enterprise, where in a year's time the money will increase to $130 You have agreed that your friend will then repay you $120, keeping $10 for...
-
Consider how you and your OS use multitasking or how other OS resources mimic real life, like cooking dinner. Discuss in a narrative form how you need to switch your attention and start and stop the...
-
Assume for a moment that in dogs, curly hair (C) is dominant to straight hair (c). Suppose you breed a homozygous curly hair male with a straight hair female. If mating occurred between the F1...
-
1. What are the important trends in the industry and general environments that Nintendo will have to keep up with in the coming years? 2+ trends identified, 2+ future competency suggestions, with...
-
At ThyssenKrupp, the data collection process was largely paper based. This made it very difficult for employees to quickly find important information in documents that were constantly being shipped...
-
Martin Wireless reported these figures in its December 31, 2020, financial statements. K i (Click the Icon to view the figures.) Compute the gross margin percentage and rate of inventory turnover for...
-
Show that every group G with identity e and such that x * x = e for all x G is abelian.
-
Is molality or molarity dependent on temperature? Explain your answer. Why is molality, and not molarity, used in the equations describing freezing-point depression and boiling-point elevation?
-
How does probability fit into the description of the atom?
-
Calculate the pH of a 5.0 10 -3 -M solution of H 2 SO 4 .
-
Suppose that noncollege-graduates earn a salary of $25,000 per year in your hometown, and that salary will never change. If you go to college for four years, foregoing four years worth of that...
-
In 2002, Donnin Company had a break-even point of \($350,000\) based on a selling price of \($7\) per unit and fixed costs of \($140,000\). In 2003, the selling price and the variable cost per unit...
-
Ewing Company estimates that variable costs will be 50% of sales, and fixed costs will total \($800,000\). The selling price of the product is $4. Instructions (a) Prepare a CVP graph, assuming...

Study smarter with the SolutionInn App



