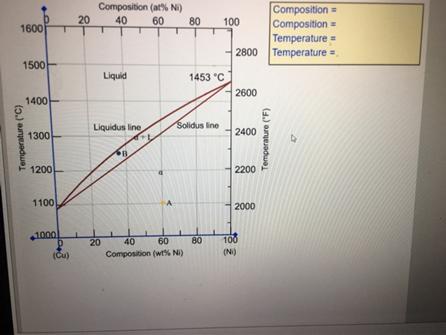
A 40 wt% Ni-60 wt% Cu alloy (Animated Figure 9.3a) is slowly cooled from 1400C (2550F)...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A 40 wt% Ni-60 wt% Cu alloy (Animated Figure 9.3a) is slowly cooled from 1400°C (2550°F) to 1150°C (2100°F). (a) At what temperature does the first solid phase form? °C (b) What is the composition of this solid phase? %wt Ni (c) At what temperature does the last of the liquid solidify? °C (d) What is the composition of this last remaining liquid phase? %wt Ni Composition (at% Ni) 20 Composition= Composition = Temperature = = 40 60 80 100 1600 2800 Temperature 1500 Liquid 1453 °C 2600 1400- Liquidus line Solidus line 2400 1300 1200 2200 1100 2000 1000 20 40 60 80 100 (Cu) Composition (wt% NI) (N) Temperature ("C) Temperature ("F) A 40 wt% Ni-60 wt% Cu alloy (Animated Figure 9.3a) is slowly cooled from 1400°C (2550°F) to 1150°C (2100°F). (a) At what temperature does the first solid phase form? °C (b) What is the composition of this solid phase? %wt Ni (c) At what temperature does the last of the liquid solidify? °C (d) What is the composition of this last remaining liquid phase? %wt Ni Composition (at% Ni) 20 Composition= Composition = Temperature = = 40 60 80 100 1600 2800 Temperature 1500 Liquid 1453 °C 2600 1400- Liquidus line Solidus line 2400 1300 1200 2200 1100 2000 1000 20 40 60 80 100 (Cu) Composition (wt% NI) (N) Temperature ("C) Temperature ("F)
Expert Answer:
Related Book For 

Applied Physics
ISBN: 978-0132109277
10th Edition
Authors: Dale ewen, Neill schurter, P. erik gundersen
Posted Date:
Students also viewed these mechanical engineering questions
-
At what temperature does the rms speed of? (a) H2 (molecular hydrogen) and (b) O2 (molecular oxygen) equals the escape speed from Earth (Table 13-2)? At what temperature does the rms speed of? (c) H2...
-
At what temperature does the rms speed of O 2 molecules equal 475. m/s?
-
At what temperature does the rms speed of O2 molecules equal 400. m/s?
-
The stockholders equity section of University Fashions is presented here. University Fashions Balance Sheet (Stockholders Equity Section) ($ in thousands) Stockholders equity: Preferred stock, $50...
-
Suppose that X ~ N(-7, 14). Find. (a) P(X 0) (b) P(X -10) (c) P(-15 X -1) (d) P(-5 X 2) (e) P(|X + 7| 8) (f) The value of X for which P(X x) = 0.75 (g) The value of x for which P(X x) = 0.27...
-
What does the McWane saga tell us about the limitations of each of the following as means of protecting the safety and health of employees?
-
An effective way to learn how companies respond to the competing pressures to be globally integrated and locally responsive is to study them in action. Referring back to Exhibit 6.3, search online...
-
Sutton Industrial Products Inc. (SIPI) is a diversified industrial-cleaner processing company. The companys Verde plant produces two products: a table cleaner and a floor cleaner from a common set of...
-
What is the difference between a & b? Why? Compare inflation for 1970s and 1980s. Any improvement in the 1990s? What might be one reason for the improvement?
-
Complete Form 941 for the 4th quarter for TCLH Industries (which is located at 202 Whitmore Avenue, Durham, NC 27701; Employer Identification #44-4444444). Assume that all necessary deposits were...
-
JetBlue Airlines Revenue (n = 8 years) Year...........................Revenue (mil) 2008...........................3,388 2009...........................3,286 2010...........................3,779...
-
All submissions to be graded for this assignment must be made using the Submit Assignment button at this posting Do not post any files in comments Seven points will be deducted from your grade if you...
-
Need to identify both critical insights and critical flaws to critique. How is biomimicry changing the world? Biomimicry is just as it sounds, mimicking our natural life on Earth in human-made...
-
Welcome(s) to the discussion of week. The following study questions are intended to illustrate the application of the concepts studied in the units. 1. What makes Baby Boomers more productive and...
-
Travel arrangement problems Angelina Morganhouse is an administrative professional for a telecommunications company. Last week her supervisor, Elena Perez, asked her to make arrangements for Ms....
-
Given a generic HashNode as a parameter // return the value in the HashNode. public static V returnValue(HashNode node) { } // Given a generic HashNode as a parameter // return the key in the...
-
For each of the business events listed in the following, (1) identify the business process for the event in a process-based approach and (2) identify the traditional accounting cycle(s) for the...
-
You are maintaining a subsidiary ledger account for Police-Training Expenditures for 2013. The following columns are used: Inventory purchases are initially recorded as expenditures. Record the...
-
1. A metal casting displaces 327 cm3 of water. Find the buoyant force of the water. 2. A piece of metal displaces 657 cm3 of water. Find the buoyant force of the water. 3. A metal casting displaces...
-
A window washer with mass 90.0 kg first climbs 45.0 m upward to the top of a building, then from the top goes down 85.0 m to the ground. (a) What is the potential energy of the window washer at the...
-
Why is a sunset red?
-
How to include a \(0-1\) indicator variable on the right-hand side of a regression, how this affects model interpretation, and give an example.
-
How to interpret the coefficient on an indicator variable in a log-linear equation.
-
How to test the equivalence of two regression equations using indicator variables.

Study smarter with the SolutionInn App



