A 41 g sample of sodium bicarbonate solution, NaHCO,, was reacted with an excess of HCI...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
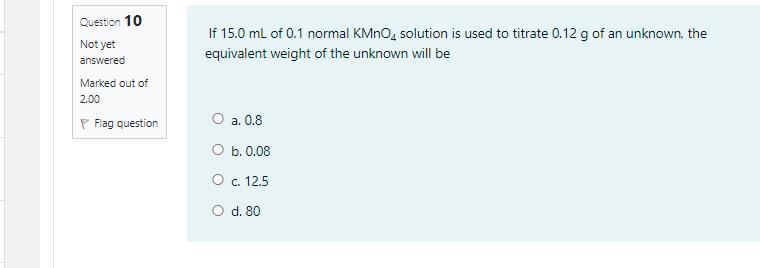
A 41 g sample of sodium bicarbonate solution, NaHCO,, was reacted with an excess of HCI as in the equation NaHCO, + HCI - NaCI + CO, + H20 If 0.06 mol of NaCl was obtained at the end of the experiment, the percentage of NaHCO3 in the sample is [Molar mass of NaHCO; = 84.01 g mol] O a. 0.146 % О ь.0.12% O c. 12.3% O d. 50 % Question 10 If 15.0 ml of 0.1 normal KMNO, solution is used to titrate 0.12 g of an unknown, the Not yet equivalent weight of the unknown will be answered Marked out of 2.00 P Flag question O a. 0.8 O b. 0.08 O c. 12.5 O d. 80 A 41 g sample of sodium bicarbonate solution, NaHCO,, was reacted with an excess of HCI as in the equation NaHCO, + HCI - NaCI + CO, + H20 If 0.06 mol of NaCl was obtained at the end of the experiment, the percentage of NaHCO3 in the sample is [Molar mass of NaHCO; = 84.01 g mol] O a. 0.146 % О ь.0.12% O c. 12.3% O d. 50 % Question 10 If 15.0 ml of 0.1 normal KMNO, solution is used to titrate 0.12 g of an unknown, the Not yet equivalent weight of the unknown will be answered Marked out of 2.00 P Flag question O a. 0.8 O b. 0.08 O c. 12.5 O d. 80
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A sample of zinc metal was reacted with an excess of hydrochloric acid. Zn(s) + 2HCl(aq) ZnCl2(aq) + H2(g) All of the zinc reacted, and the hydrogen gas was collected over water at 17oC; the...
-
A sample of sodium peroxide, Na2O2, was reacted with an excess of water. 2Na2O2(s) + 2H2O(l ) 4NaOH(aq) + O2(g) All of the sodium peroxide reacted, and the oxygen was collected over water at 21oC....
-
A 15.5 g sample of sodium carbonate is added to a solution of acetic acid weighing 19.7 g. The two substances react, releasing carbon dioxide gas to the atmosphere. After reaction, the contents of...
-
Consider the case of the Cisco Fatty. Who was wrong? Advise how a firm might best handle this kind of online commentary.
-
Identify the factors expanding the international capital market. What is meant by the term securitization?
-
A partial list of the accounts and ending account balances taken from the post closing trial balance of the Jordan Corporation on December 31, 2007 is shown as follows: Account Title Amount Retained...
-
John and Jennifer Margeson entered into a contract to sell a weight-loss franchise business called Inches-A-Weigh to Theresa Artis. The parties memorialized their agreement in an Asset Purchase...
-
Error and Change in Estimate Depreciation Tarkington Co. purchased a machine on January 1, 2007, for $440,000. At that time it was estimated that the machine would have a 10-year life and no salvage...
-
What payment, made at the end of each year for 13 years, will accumulate to $11,300 at 11% compounded monthly? The required annual payment is $ (Round the final answer to the nearest cent as needed....
-
On December 31, 2017, Jen & Mink Clothing (J&M) performed the inventory count and determined the year-end ending inventory value to be $75,500. It is now January 8, 2018, and you have been asked to...
-
If one of your herds that you visit is averaging 82/lb/day of milk, the farmer is receiving $21.50/cwt for the milk, the cows are eating an average of 52 lb/day of DM that cost $0.1250/lb, calculate...
-
What is 'tailoring the process' in context of project management?
-
The term structure of interest rates is flat at 8%. You want to immunize a liability of $1,000 maturing in 7 years. To do so you may invest in a combination of the following a perpetuity with a...
-
A 2.0-mm-diameter, 50-cm-long copper wire carries a 2.5 A current. What is the potential difference between the ends of the wire? (mV)
-
Prepare Journal Entry ? Accounts Cash Accounts Receivable Supplies Equipment Accumulated Depreciation Salaries Payable Common Stock Retained Earnings Totals Debits Credits $ 13,500 6,700 2,700 16,500...
-
The manufacturing costs of Ackerman Industries for the first three months of the year follow: Total Costs Units Produced January $60,280 1,440 units February 56,160 845 March 87,360 2,145 Using the...
-
4. Given an array, A, of n integers, give an O(n)-time algorithm that finds the longest subarray of A such that all the numbers in that subarray are in sorted order. Your algorithm outputs two...
-
Prove the following D,(cos x) = - sin x (Hint: Apply the identity cos(A + B) = cos A cos B sin A sin B)
-
Magnesite (magnesium carbonate, MgCO3) is a common magnesium mineral. From the solubility product constant (Table 17.1), find the solubility of magnesium carbonate in grams per liter of water. TABLE...
-
Curium was first synthesized by bombarding an element with alpha particles. The products were curium-242 and a neutron. What was the target element?
-
Describe any thermochemical (heat of reaction) evidence for the Arrhenius concept.
-
See Table 2.5 showing financial statement data and stock price data for Mydeco Corp. Was Mydeco able to improve its ROIC in 2023 relative to what it was in 2019? TABLE 2.5 2019-2023 Financial...
-
You have an investment opportunity in Japan. It requires an investment of \($0.98\) million today and will produce a cash flow of 107 million in one year with no risk. Suppose the risk-free interest...
-
Motor Company is considering offering a \($1,600\) rebate on its minivan, lowering the vehicles price from \($29,000\) to \($27,400.\) The marketing group estimates that this rebate will increase...

Study smarter with the SolutionInn App


