If the theoretical yield for a reaction was 131 grams and I actually made 112 grams...
Fantastic news! We've Found the answer you've been seeking!
Question:
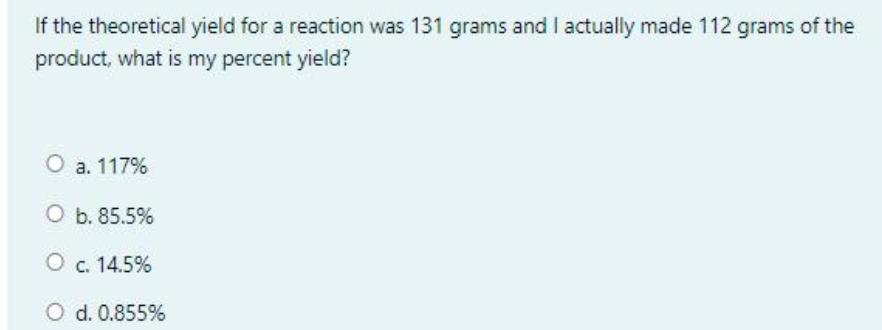

Transcribed Image Text:
If the theoretical yield for a reaction was 131 grams and I actually made 112 grams of the product, what is my percent yield? O a. 117% O b. 85.5% O c. 14.5% O d. 0.855% Question 12 The gas produced from the reaction of NaHCO; and HCI is Not yet answered Marked out of O a. H2O 2.00 P Flag question O b. NaCI O c. CO2 O d. Na,CO; If the theoretical yield for a reaction was 131 grams and I actually made 112 grams of the product, what is my percent yield? O a. 117% O b. 85.5% O c. 14.5% O d. 0.855% Question 12 The gas produced from the reaction of NaHCO; and HCI is Not yet answered Marked out of O a. H2O 2.00 P Flag question O b. NaCI O c. CO2 O d. Na,CO;
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following statements is true of embargoes? Select one: O a. An embargo may be placed on one or a few goods or may completely ban trade in all goods. O b. An embargo is the least...
-
Muscat audit firm is conducting audit for Suhail Bahwan Group since 15 days. Muscat audit firm needs more time to continue the audit because of the size and profile of Bahwan group. The audit firm...
-
Which of the following best describes an ad valorem tax? Select one: a). A tax levied as a percentage of the price of a good purchased b). A constant tax levied on each unit produced c). A fixed tax...
-
The following is a symbolic microprogram for an instruction in the computer defined in Sec. 7-3. a. Specify the operation performed when the instruction is executed. b. Convert the four...
-
A local supermarket has contracted for floor cleaning and polishing to be done each evening by a small work crew using mechanized equipment. After the crew has performed the service for several weeks...
-
_______________ allow organizations to measure their performance in certain areassuch as failure rates, availability, and reliability and compare them over time or with other organizations. A....
-
Journalize the following transactions for Amazing Audio, Inc., that occurred during the month of November. Amazing Audios cost of inventory is 65% of the sales price. Nov 3 Sold $1,600 of merchandise...
-
Diana Capriati, a Canadian resident for income tax purposes, has the following income for 2010 and 2011: REQUIRED Based upon the information provided, determine the maximum RRSP contribution that...
-
Dror purchases a stock at $40, receives a $3.00 dividend, and later sells the stock for $44. What is the holding period return for this investment?
-
AP Vianne is the management accountant for Divvy, a bike-sharing service in Chicago. She is trying to plan for summer activity by using the most recent ridership information provided. She needs to...
-
Crystal A diffracts from (1 1 1) and (2 0 0) planes but not from (110) plane, while the crystal B diffracts from (110) and (2 0 0) planes but not from the (111) plane. From the above, we may conclude...
-
Why do companies divest assets?
-
In the mortgage industry, more and more nonconforming loans were purchased, pooled, and segmented into tranches of different grades of risk and then sold to investors as __________ securities.
-
The Commodities Futures Modernization Act of 2000 and the repeal of the Glass-Steagall Act in 1999: (a) Effectively ended the separation of commercial banking from investment banking. (b) Ended the...
-
Explain the reset clause in the pay-option ARM loan agreement.
-
In small groups or in pairs, and based upon the current economic environment, research and discuss top CFO priorities.
-
Date March 1 Activities Beginning inventory March 5 Purchase March 9 Sales March 18 Purchase March 25 Purchase March 29 Sales Totals 70 units 210 units Units Acquired at Cost @$50.40 per unit @...
-
-x/2 x/4 If A = -x/2 and A-1 =6 then x equals
-
Which of the following species has the most unpaired electrons? S+, S, or S-. Explain how you arrive at your answer.
-
Group the following electron configurations in pairs that would represent similar chemical properties of their atoms: (a) 1s22s22p5 (b) 1s22s1 (c) 1s22s22p6 (d) 1s22s22p63s23p5 (e) 1s22s22p63s23p64s1...
-
A 0.86 percent by mass solution of NaCl is called "physiological saline" because its osmotic pressure is equal to that of the solution in blood cells. Calculate the osmotic pressure of this solution...
-
Cindy Belton opened a law office, Cindy Belton, Attorney at Law, on July 1, 2008. On July 31, the balance sheet showed Cash \($4,000,\) Accounts Receivable \($1,500,\) Supplies \($500,\) Office...
-
The actual financial statements of PepsiCo, as presented in the companys 2005 Annual Report, are contained in Appendix A (at the back of the textbook). Instructions Refer to PepsiCos financial...
-
PepsiCos financial statements are presented in Appendix A. The Coca-Cola Companys financial statements are presented in Appendix B. Instructions (a) Based on the information contained in these...

Study smarter with the SolutionInn App



