A 46.91 mL solution of weak acid is titrated with 0.429 M NaOH. It requires 34.58...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
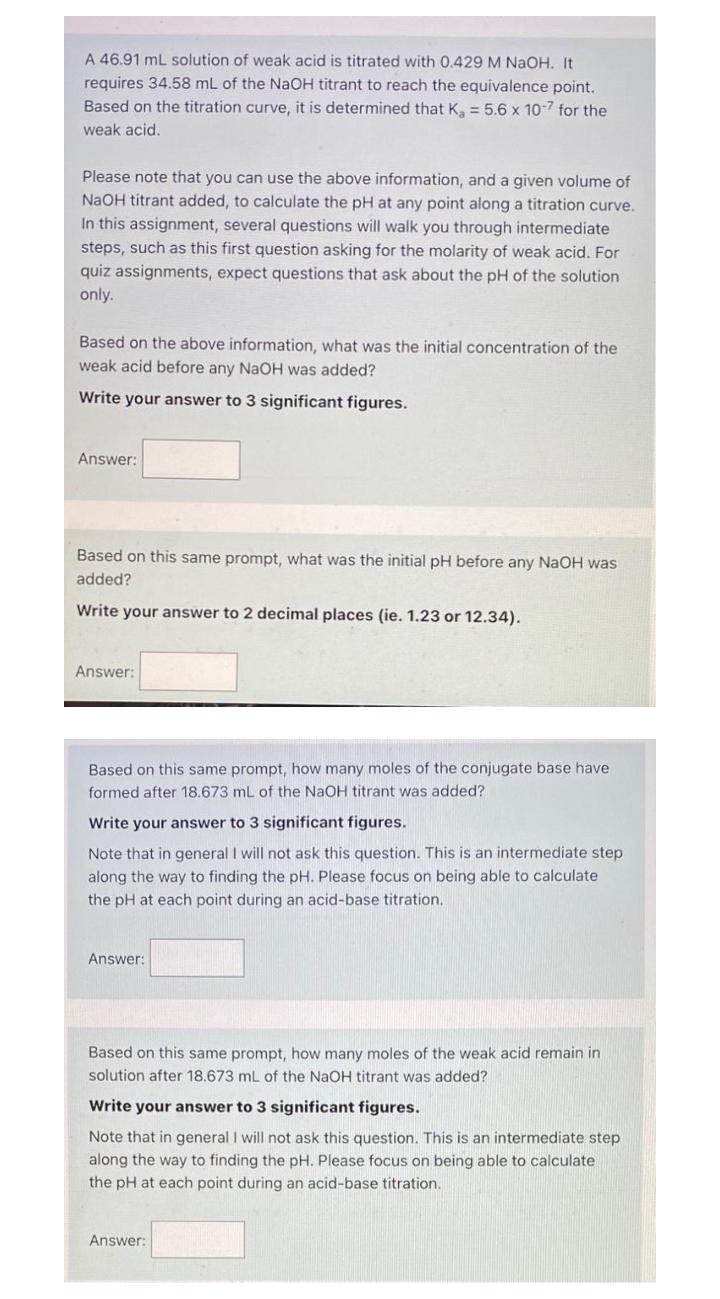
A 46.91 mL solution of weak acid is titrated with 0.429 M NaOH. It requires 34.58 mL of the NaOH titrant to reach the equivalence point. Based on the titration curve, it is determined that K = 5.6 x 10-7 for the weak acid. Please note that you can use the above information, and a given volume of NaOH titrant added, to calculate the pH at any point along a titration curve. In this assignment, several questions will walk you through intermediate steps, such as this first question asking for the molarity of weak acid. For quiz assignments, expect questions that ask about the pH of the solution only. Based on the above information, what was the initial concentration of the weak acid before any NaOH was added? Write your answer to 3 significant figures. Answer: Based on this same prompt, what was the initial pH before any NaOH was added? Write your answer to 2 decimal places (ie. 1.23 or 12.34). Answer: Based on this same prompt, how many moles of the conjugate base have formed after 18.673 mL of the NaOH titrant was added? Write your answer to 3 significant figures. Note that in general I will not ask this question. This is an intermediate step along the way to finding the pH. Please focus on being able to calculate the pH at each point during an acid-base titration. Answer: Based on this same prompt, how many moles of the weak acid remain in solution after 18.673 mL of the NaOH titrant was added? Write your answer to 3 significant figures. Note that in general I will not ask this question. This is an intermediate step along the way to finding the pH. Please focus on being able to calculate the pH at each point during an acid-base titration. Answer: A 46.91 mL solution of weak acid is titrated with 0.429 M NaOH. It requires 34.58 mL of the NaOH titrant to reach the equivalence point. Based on the titration curve, it is determined that K = 5.6 x 10-7 for the weak acid. Please note that you can use the above information, and a given volume of NaOH titrant added, to calculate the pH at any point along a titration curve. In this assignment, several questions will walk you through intermediate steps, such as this first question asking for the molarity of weak acid. For quiz assignments, expect questions that ask about the pH of the solution only. Based on the above information, what was the initial concentration of the weak acid before any NaOH was added? Write your answer to 3 significant figures. Answer: Based on this same prompt, what was the initial pH before any NaOH was added? Write your answer to 2 decimal places (ie. 1.23 or 12.34). Answer: Based on this same prompt, how many moles of the conjugate base have formed after 18.673 mL of the NaOH titrant was added? Write your answer to 3 significant figures. Note that in general I will not ask this question. This is an intermediate step along the way to finding the pH. Please focus on being able to calculate the pH at each point during an acid-base titration. Answer: Based on this same prompt, how many moles of the weak acid remain in solution after 18.673 mL of the NaOH titrant was added? Write your answer to 3 significant figures. Note that in general I will not ask this question. This is an intermediate step along the way to finding the pH. Please focus on being able to calculate the pH at each point during an acid-base titration. Answer:
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Posted Date:
Students also viewed these chemistry questions
-
A weak monoprotic acid is titrated with 0.100 M NaOH. It requires 50.0 mL of the NaOH solution to reach the equivalence point. After 25.0 mL of base is added, the pH of the solution is 3.62. Estimate...
-
Note that you can use the Black-Scholes formula to calculate the call option premium as a percentage of the exercise price in terms of S/X: where Implement this in a spreadsheet. C=SN(d))-Xe"T...
-
An acid is titrated with NaOH. The following beakers are illustrations of the contents of the beaker at various times during the titration. These are presented out of order. Note: Counter-ions and...
-
On July 31, 2019, the balances of the accounts appearing in the ledger of Serbian Interiors Company, a furniture wholesaler, are as follows: Prepare the July 31, 2019, closing entries for Serbian...
-
Would it be a violation of equal protection for a state to impose a higher tax on out-of-state companies doing business in the state than it imposes on in-state companies if the only reason for the...
-
An unlevered firm has a cost of capital of 16% and earnings before interest and taxes of $225,000. A levered firm with the same operations and assets has both a book value and a face value of debt of...
-
How is the name of a use case determined?
-
Prescott, Inc. manufactures bookcases and uses an activity-based costing system. Prescotts activity areas and related data follow: Prescott produced two styles of bookcases in October: the standard...
-
Selected information from the Iowa Instruments accounting records for April follows: Materials Inventory Debit Credit BB ( 4 / 1 ) 2 7 , 0 0 0 2 1 1 , 0 0 0 1 7 6 , 0 0 0 Work - in - Process...
-
Ellipses Corp is a small business that operates in Herndon, VA. The company is located at10 Period Lane, Herndon, VA 20170. Its federal Employer Identification Number is 77-7777777, and its...
-
Stock Exchange Presentation Task: You are a senior execute at an ASX-listed company. If it is helpful, you may nominate a specific ASX-listed company to represent. The CEO of your company is...
-
If two planets have the same mass, but \(A\) has twice the radius of \(B\), determine what the ratio of the accelerations of gravity is worth \(g_{A} / g_{B}\).
-
Show that if the magnitudes of the sum and difference between two vectors are equal, then the vectors are perpendicular to each other.
-
Show that the force field expressed in spherical coordinates \(\mathbf{F}=f(r, \theta) \mathbf{r}\) is conservative if and only if the function \(f\) is independent of \(\theta\).
-
The equation of motion of a material point is expressed by the relation \(x(t)=\alpha t^{3}-\beta t^{2}-\gamma\), with the constants \(\alpha, \beta, \gamma\) real positive. Determine velocity and...
-
A particle moves in uniformly accelerated motion on a straight line. After \(t_{1}=4 \mathrm{~s}\) it has traveled \(60 \mathrm{~m}\) and has a velocity \(v_{1}=33 \mathrm{~m} / \mathrm{s}\)....
-
Required Prepare Schedule of Cost of Goods Manufactured and Sold At December 31,2016 , the end of its fiscal year. At December 31,2016, the end of its fiscal year, Lederman Manufacturing Corporation...
-
A non-charmed baryon has strangeness S = 2 and electric charge Q = 0. What are the possible values of its isospin I and of its third component I z ? What is it usually called if I = 1/2?
-
(a) Determine the number of calcium ions in the chemical formula of the mineral hdystonite, Ca x Zn(Si 2 O 7 ). (b) Determine the number of hydroxide ions in the chemical formula of the mineral...
-
In the experiment shown schematically below, a beam of neutral atoms is passed through a magnetic field. Atoms that have unpaired electrons are deflected in different directions in the magnetic field...
-
State whether each of the statements below is true or false. Justify your answer in each case. (a) Electrolyte solutions conduct electricity because electrons are moving through the solution. (b) If...
-
Some people argue that the government should not intervene in the case of a market failure because the government itself is inefficient and will simply create new problems to replace the ones it is...
-
Consider each of the following issues and discuss whether you support Theory X, Theory Y, neither theory, or some combination of them. Issue Theory X Theory Y Whether a person is healthy or sick...
-
Looking at Medicaids traditional eligibility rules, you will notice numerous value/policy judgmentspregnant women and children are favored over childless adults, the medically needy are favored over...

Study smarter with the SolutionInn App



