A concentrated brine solution (aqueous sodium chloride) contains magnesium as an impurity. A membrane with surface...
Fantastic news! We've Found the answer you've been seeking!
Question:
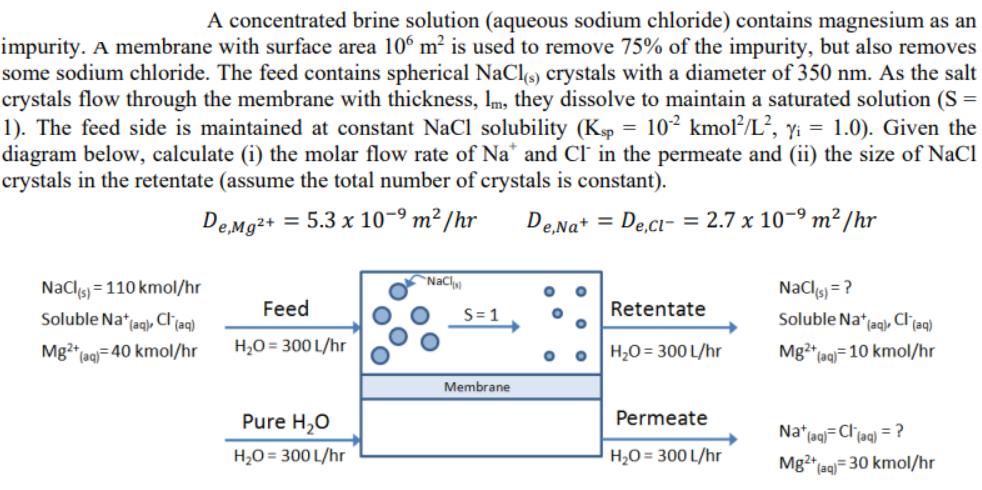
Transcribed Image Text:
A concentrated brine solution (aqueous sodium chloride) contains magnesium as an impurity. A membrane with surface area 106 m² is used to remove 75% of the impurity, but also removes some sodium chloride. The feed contains spherical NaCl(s) crystals with a diameter of 350 nm. As the salt crystals flow through the membrane with thickness, Im, they dissolve to maintain a saturated solution (S = 1). The feed side is maintained at constant NaCl solubility (Ksp = 102 kmol/L², y₁ = 1.0). Given the diagram below, calculate (i) the molar flow rate of Na* and CI in the permeate and (ii) the size of NaCl crystals in the retentate (assume the total number of crystals is constant). De,Mg²+ = 5.3 x 10-⁹ m²/hr De,Na+ = De,Cl- = 2.7 x 10-⁹ m²/hr NaCl(s) 110 kmol/hr Soluble Na+ (aq), Cl(aq) Mg2+ (aq) 40 kmol/hr Feed H₂O = 300 L/hr Pure H₂O H₂O = 300 L/hr NaCl S=1 Membrane O O O Retentate H₂O=300L/hr Permeate H₂O = 300 L/hr NaCl(s) = ? Soluble Nat (aq), Cl(aq) Mg2+ (aq) 10 kmol/hr Na+ (aq)=Cl(aq) = ? Mg2+ (aq)= 30 kmol/hr A concentrated brine solution (aqueous sodium chloride) contains magnesium as an impurity. A membrane with surface area 106 m² is used to remove 75% of the impurity, but also removes some sodium chloride. The feed contains spherical NaCl(s) crystals with a diameter of 350 nm. As the salt crystals flow through the membrane with thickness, Im, they dissolve to maintain a saturated solution (S = 1). The feed side is maintained at constant NaCl solubility (Ksp = 102 kmol/L², y₁ = 1.0). Given the diagram below, calculate (i) the molar flow rate of Na* and CI in the permeate and (ii) the size of NaCl crystals in the retentate (assume the total number of crystals is constant). De,Mg²+ = 5.3 x 10-⁹ m²/hr De,Na+ = De,Cl- = 2.7 x 10-⁹ m²/hr NaCl(s) 110 kmol/hr Soluble Na+ (aq), Cl(aq) Mg2+ (aq) 40 kmol/hr Feed H₂O = 300 L/hr Pure H₂O H₂O = 300 L/hr NaCl S=1 Membrane O O O Retentate H₂O=300L/hr Permeate H₂O = 300 L/hr NaCl(s) = ? Soluble Nat (aq), Cl(aq) Mg2+ (aq) 10 kmol/hr Na+ (aq)=Cl(aq) = ? Mg2+ (aq)= 30 kmol/hr
Expert Answer:

Related Book For 

Smith and Roberson Business Law
ISBN: 978-0538473637
15th Edition
Authors: Richard A. Mann, Barry S. Roberts
Posted Date:
Students also viewed these chemical engineering questions
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
A cylinder 1.00 m tall with inside diameter 0.120 m is used to hold propane gas (molar mass 44.1 g/mol) for use in a barbecue. It is initially filled with gas until the gauge pressure is 1.30 X 106...
-
A Friedel-Crafts acylation of anisole with acetyl chloride affords a binary product with a total mass of 1.91 grams. This product is analyzed by gas chromatograph. The chromatogram shows two product...
-
1. Think about which exercise (part a or part b)may be completed by multiplying by 6. Now complete each exercise. a. Solve: x/6 - 5/3 = 2 b. Subtract: x/6 - 5/3 2. 19/53 = 353x / 1431 + 23/27 3. The...
-
Find the general solution for each differential equation. dy dx = 4x + 6x5
-
If you want to maintain a potential difference of \(6000 \mathrm{~V}\) between the plates of a parallel-plate capacitor, what is the minimum value of the plate separation if the space between the...
-
Rainey Enterprises loaned $20,000 to Small Co. on June 1, 2016, for one year at 6 percent interest. Required a. Record these general journal entries for Rainey Enterprises: (1) The loan to Small Co....
-
The DD-AA model predicts that a permanent increase in foreign money demand will lead to an improvement in the domestic current account. True/False/Uncertain, explain and support your answer with ONE...
-
An army division in Iraq has five troop encampments in the desert, and the division leaders want to determine the best location for a supply depot to serve the camps. The (x, y) coordinates (in...
-
Hi! How can I make a JAVA class given by the following UML? ________________________________________________________ - author : String - title : String - cover : String - pages : int - copyright :...
-
Question: How and why did piracy affect European empires in the Atlantic world from the late sixteenth to the early eighteenth centuries? - In your response, you might like to consider the role of...
-
What is the background, facts and conclusions, comments from the judge about this case? A brief summary with facts. Simpsons limited v canadian union of brewery, flour, Cereal, Soft Drink and...
-
Consider a U.S. Corporate bond that pays semi-annual coupons with settlement of May 12, 2018. The bond matures on March 1, 2046. The bond's coupon rate is 3% and has a yield of 3%. What is the bond's...
-
The Quarantine Corporation began operations in March 2024. The following is a condensed balance sheet as of March 31, 2024. The owners did not take any distributions from the business in March. Cash...
-
An amount of AED 24900 is deposited to a savings account for 50 weeks at a simple interest rate of 3.1% per annum. How much interest did your money earn at the end of the period
-
The Greek word for actor is "hypokrite," the source of the English word "hypocrite." Look the term up in the dictionary. Is role-playing in real life hypocritical? Are actors not to be trusted...
-
A statistical study shows that the fraction of television sets of a certain brand that are still in service after x years is given by f (x) = e-0.15x. (a) What fraction of the sets are still in...
-
Explain the international dimensions of antitrust law, securities regulation, the protection of intellectual property, and employment discrimination.
-
Rebecca owes Lewis $2,500 due on November 1. On August 15, Lewis assigns this right for value received to Julia, who gives notice on September 10 of the assignment to Rebecca. On August 25, Lewis...
-
Civil Code 1719, subdivision (a) provides in part that any person who draws a check that is dishonored due to insufficient funds shall be liable to the payee for the amount owing upon the check and...
-
A significance level of 0.05 indicates that the probability of making a type I error is 0.05.
-
A handy mnemonic for interpreting the P-value in a hypothesis test is this: If the P (value) is low, then the null must go.
-
In testing a claim about a population mean, a larger z test statistic always results in a larger P-value. Decide whether the statement makes sense (or is clearly true) or does not make sense (or is...

Study smarter with the SolutionInn App


