A cylinder of volume 0.270 m contains 11.1 mol of neon gas at 19.8C. Assume neon...
Fantastic news! We've Found the answer you've been seeking!
Question:
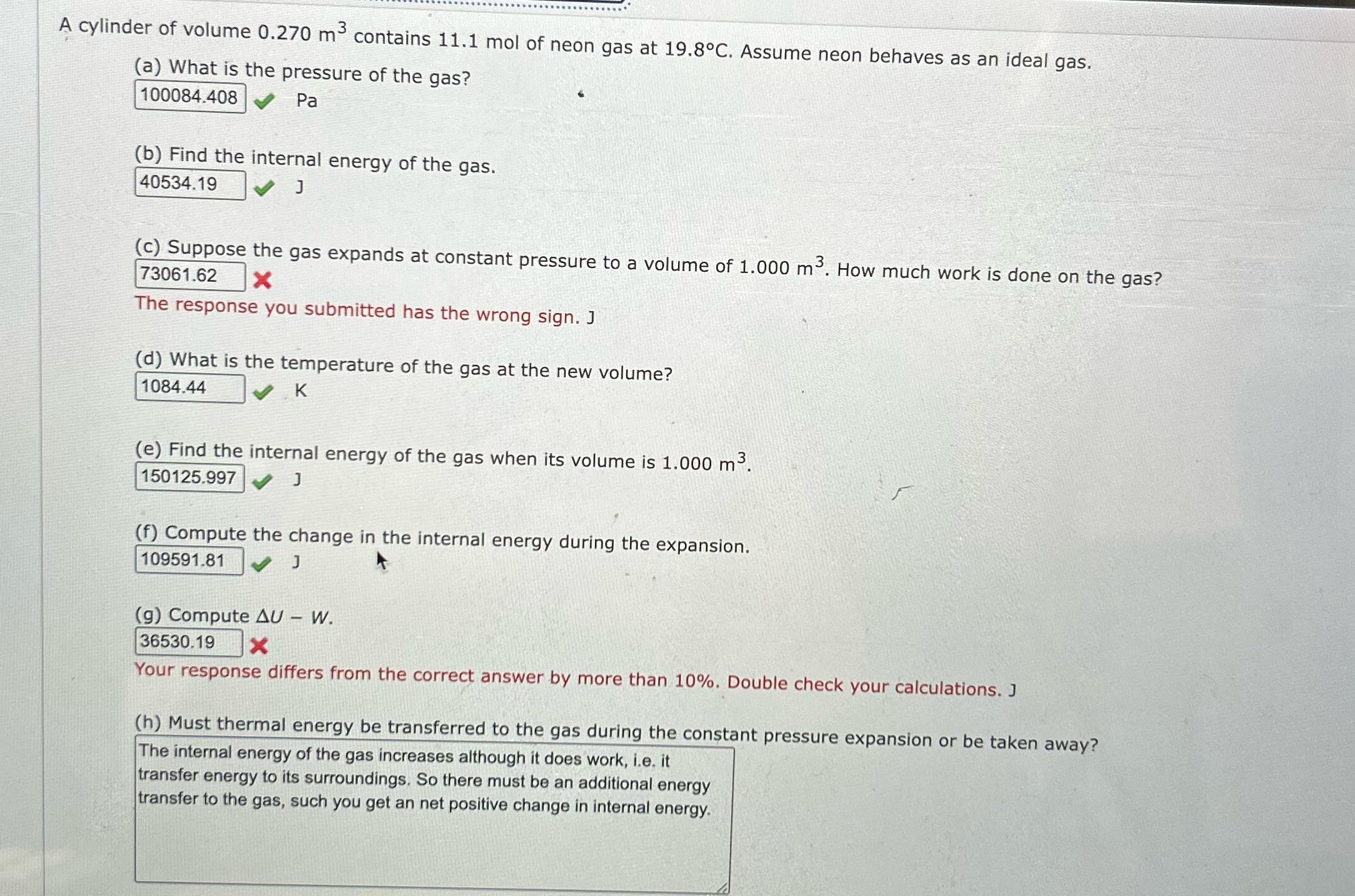
Transcribed Image Text:
A cylinder of volume 0.270 m³ contains 11.1 mol of neon gas at 19.8°C. Assume neon behaves as an ideal gas. (a) What is the pressure of the gas? 100084.408 Pa (b) Find the internal energy of the gas. 40534.19 J (c) Suppose the gas expands at constant pressure to a volume of 1.000 m³. How much work is done on the gas? 73061.62 X The response you submitted has the wrong sign. J (d) What is the temperature of the gas at the new volume? 1084.44 K (e) Find the internal energy of the gas when its volume is 1.000 m³. 150125.997✔✔ J (f) Compute the change in the internal energy during the expansion. 109591.81 ✔ J (g) Compute AU - W. 36530.19 Your response differs from the correct answer by more than 10%. Double check your calculations. J (h) Must thermal energy be transferred to the gas during the constant pressure expansion or be taken away? The internal energy of the gas increases although it does work, i.e. it transfer energy to its surroundings. So there must be an additional energy transfer to the gas, such you get an net positive change in internal energy. A cylinder of volume 0.270 m³ contains 11.1 mol of neon gas at 19.8°C. Assume neon behaves as an ideal gas. (a) What is the pressure of the gas? 100084.408 Pa (b) Find the internal energy of the gas. 40534.19 J (c) Suppose the gas expands at constant pressure to a volume of 1.000 m³. How much work is done on the gas? 73061.62 X The response you submitted has the wrong sign. J (d) What is the temperature of the gas at the new volume? 1084.44 K (e) Find the internal energy of the gas when its volume is 1.000 m³. 150125.997✔✔ J (f) Compute the change in the internal energy during the expansion. 109591.81 ✔ J (g) Compute AU - W. 36530.19 Your response differs from the correct answer by more than 10%. Double check your calculations. J (h) Must thermal energy be transferred to the gas during the constant pressure expansion or be taken away? The internal energy of the gas increases although it does work, i.e. it transfer energy to its surroundings. So there must be an additional energy transfer to the gas, such you get an net positive change in internal energy.
Expert Answer:
Answer rating: 100% (QA)
Heres the corrected solution with explanations a Pressure of the gas We can use the ideal gas law PV ... View the full answer

Related Book For 

Fundamentals of Thermodynamics
ISBN: 978-0471152323
6th edition
Authors: Richard E. Sonntag, Claus Borgnakke, Gordon J. Van Wylen
Posted Date:
Students also viewed these physics questions
-
Using JAVA coding: How do I create program that will allow the user to enter the degrees in Fahrenheit and output the degrees in Celsius? This program will continue to run until the user enters a 0....
-
bsolutely and you know its so true is i found that in my own life as well and I think Tony puts it perfectly he says that most people rise to the expectations of their peer group you know they did a...
-
If a company had a beginning retained earnings balance of $100,000, net income for the year of $20,000, and paid dividends of $5,000, what would the ending retained earnings balance be?
-
In Simpleland, there are only two risky assets in the market, A and B. The information and CAPM estimates of A and B are shown in the following table: A 100 Million 0.09 Market Capitalisation ($)...
-
Using a spreadsheet, prepare a cash budget for New Tech for the next year (January through December) based on the following informations. Show you calculation in detail New Tech Oct Nov Dec Sales...
-
Frame the initial issue(s) arising from the following facts. What additional information would be helpful? a. Taxpayer purchases a ticket for the New Jersey lottery. He won $ 1,000. b. Taxpayer paid...
-
Visit SunGards web site, http://www.sungard.com, and research its recovery services offered for the following classes: High Availability, System Recovery, and End-User Recover. Write a report of your...
-
1. How does the concept of segmentation pricing relate to Washburn Guitars four different price points? 2. How does Washburns four different price points reflect customer wants and needs? 3. Are...
-
18. Find the value of tan 32 24' to four places. A. 6.328 B. 0.6672 C. 0.6346 D. 0.6458
-
Write a function: string solution (string &S, string &T); that, given two strings S and T consisting of N and M characters, respectively, determines whether string T can be obtained from string S by...
-
Use the following results from a test for marijuana use, which is provided by a certain drug testing company. Among 149 subjects with positive test results, there are 22 false positive results; among...
-
On January 1, Walker Inc. acquired equipment for $80,000. The expected useful life is 10 years and the residual value is $1,600. Total service hours for the equipment are estimated to be 20,000 while...
-
A and B are partners sharing profit and losses as 2:1. On 1st April 1997 they admit C as a partner 1/5th share who pays Rs.4,500 as goodwill privately. On 1st April 1998 they take D as a partner for...
-
Machinery purchased for $47,800 by Flounder Corp. on January 1, 2018, was originally estimated to have an 8-year useful life with a residual value of $3,000. Depreciation has been entered for five...
-
Which of the following is associated with a corporate level strategy? a.To expand, grow, and prosper at a global level b.It is directed at operational effectiveness c.Develop specific business units...
-
Oakmont Company has an opportunity to manufacture and sell a new product for a four-year period. The company's discount rate is 15%. After careful study, Oakmont estimated the following costs and...
-
Natalie had a very busy December. At the end of the month, after journalizing and posting the December transactions and adjus entries, Natalie prepared the following adjusted trial balance. COOKIE...
-
Linda Lopez opened a beauty studio, Lindas Salon, on January 2, 2011. The salon also sells beauty supplies. In January 2012, Lopez realized she had never filed any tax reports for her business and...
-
Two flows of air both at 200 kPa; one has 1 kg/s at 400 K and the other has 2 kg/s at 290 K. The two flows are mixed together in an insulated box to produce a single exit flow at 200 kPa. Find the...
-
Air leaves a compressor in a pipe with a stagnation temperature and pressure of 150C, 300 kPa, and a velocity of 125 m/s. The pipe has a cross-sectional area of 0.02 m2. Determine the static...
-
Consider the combustion of hydrogen with pure oxygen in a stoichiometric ratio under steady flow adiabatic conditions. The reactants enter separately at 298 K, 100 kPa and the product(s) exit at a...
-
Leeds Architectural Consultants began operations on January 2. The following activity was recorded in the company's Work in Process account for the first month of operations: Leeds Architectural...
-
Estimated cost and operating data for three companies for the upcoming year follow: Predetermined overhead rates are computed using the following bases in the three companies: Required: 1. Compute...
-
Dillon Products manufactures various machined parts to customer specifications. The company uses a job-order costing system and applies overhead cost to jobs on the basis of machine-hours. At the...

Study smarter with the SolutionInn App


