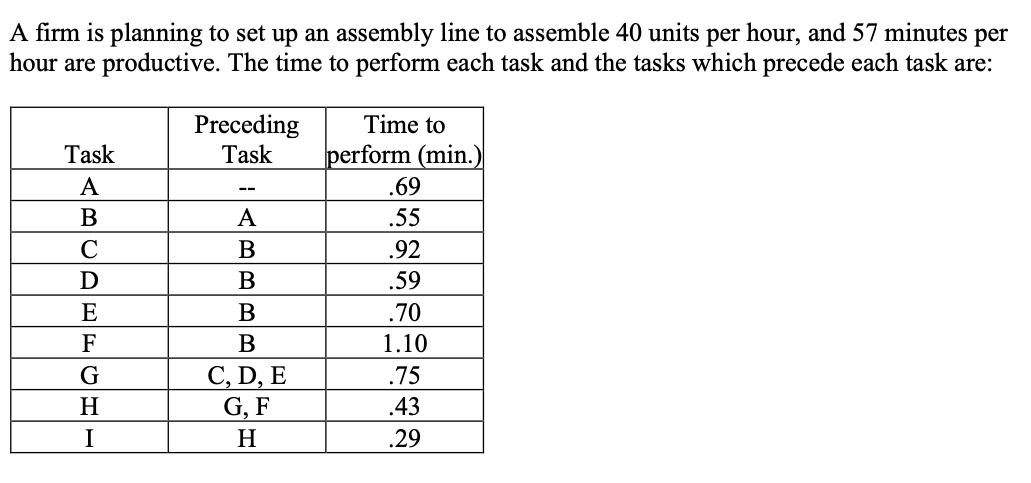
A firm is planning to set up an assembly line to assemble 40 units per hour,...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
A firm is planning to set up an assembly line to assemble 40 units per hour, and 57 minutes per hour are productive. The time to perform each task and the tasks which precede each task are: Task A B C D E F G H I Preceding Task -- A B B B B C, D, E G, F H Time to perform (min.) .69 .55 .92 .59 .70 1.10 .75 .43 .29 Q1. (5 p.) Draw a network diagram of precedence relationships. Q2. (5 p.) Compute the required cycle time per unit in minutes. Q3. (5 p.) Compute the theoretical minimum number of workstations required to produce 40 units per hour. Q4. (14 p.) Balance this line using the ranked positional weight rule. Q5. (5 p.) What is the efficiency of the line obtained in part d? A firm is planning to set up an assembly line to assemble 40 units per hour, and 57 minutes per hour are productive. The time to perform each task and the tasks which precede each task are: Task A B C D E F G H I Preceding Task -- A B B B B C, D, E G, F H Time to perform (min.) .69 .55 .92 .59 .70 1.10 .75 .43 .29 Q1. (5 p.) Draw a network diagram of precedence relationships. Q2. (5 p.) Compute the required cycle time per unit in minutes. Q3. (5 p.) Compute the theoretical minimum number of workstations required to produce 40 units per hour. Q4. (14 p.) Balance this line using the ranked positional weight rule. Q5. (5 p.) What is the efficiency of the line obtained in part d?
Expert Answer:
Answer rating: 100% (QA)
Answer Q1 Network Diagram of Precedence Relationships A 069 B C 092 D E F 110 G 075 F H 043 I 029 Q2 ... View the full answer

Related Book For 

1014 Practice Questions For The New GRE
ISBN: 9780375429682
2nd Edition
Authors: The Princeton Review
Posted Date:
Students also viewed these general management questions
-
An organization offers a program designed to increase the level of comprehension achieved by students when reading technical material quickly. Each member of a random sample of 10 students was given...
-
A portfolio manager holds a portfolio that mimics the ASX 200 Stock Index. The ASX 200 started the year at 5,000 and is currently at 6,000 points. The manager's fund was valued at $265 million at the...
-
Consider the Cobb-Douglas production function f(x, y) = 300x0.85, 0.15 When x = 4000 and y = 800, find the following. (Round your answers to two decimal places.) (a) the marginal productivity of...
-
A 120-lb pilot flies a jet trainer in a half vertical loop of 3600-ft radius so that the speed of the trainer decreases at a constant rate. Knowing that the pilot??s apparent weights at points A and...
-
A new police car costs the Bay City Police Department $26,000. The annual maintenance cost for a car depends on the age of the car at the beginning of the year. (All cars accumulate approximately the...
-
An oil preheater consists of a single tube of \(10-\mathrm{mm}\) diameter and 5-m length, with its surface maintained at \(180^{\circ} \mathrm{C}\) by swirling combustion gases. The engine oil (new)...
-
At year-end, the price-earnings ratio of General Motors was 11.3, and the price-earnings ratio of Microsoft was 28.14. Which company did the stock market favor? Explain.
-
In Thunder Bay, a public survey was done that suggests that people are generally supportive of using automated, AI-based video analysis software [1]. Yet, elsewhere in Canada, such software has come...
-
Sue and Dave always spend their evenings together, either cycling or playing computer games. On an evening with good weather, the probability that Sue prefers cycling is 0.7 and for Dave it is 0.4....
-
1 How are CoinBase competing with other crypto exchanges such as Binance, crypto.com,or FTX? Question 2 If you were hired as a summer intern by CoinBase in the strategy department, what key...
-
Gama Ltd. has to select a capital project between Project A and Project B. The details of both the projects are given below: Cash Flows Year Project A Project B O -2000 -2000 1 1500 150 2 700 850 3...
-
A pressurized container produced from cast iron is closed and sealed at the top with a circular plate cover made of steel. A total of 16 low-carbon martensitic Q&T steel bolts are used to join the...
-
In this assessment task students are required to develop a digital media strategy to provide information and promote a community service organisation to clients which could include: registering a...
-
C5: Suppose Bethany is putting money from her year-end bonus into a retirement account. Each year she saves $10,000 and can earn 10% on the money. If she saves that much each year for 40 years, how...
-
Baker LLC has $10,000 in total assets. ROA is 8% and the firm's tax rate is 20%. The firm paid interest expense of $100. What is Baker's TIE ratio?
-
Noah Collins wants to buy a jet that currently sells for $3.4 million. Its price is increasing at an annual rate of 2.3 percent. If he has already saved $2.5 million as of today, what annual rate of...
-
The text defined intrinsic value as the value of an asset given a hypothetically complete understanding of the assets investment characteristics. Discuss why hypothetically is included in the...
-
If the volume of a cube is 125, then the total area of 2 of its faces is 20 25 40 50 75
-
In square ABCE, AB = 4. Quantity A is greater. Quantity B is greater. The two quantities are equal. The relationship cannot be determined from the information given. A B E 60 C 60 60 D
-
Triangle ACE is equilateral with side lengths of 8. Points B and D are the midpoints of line segments AC and CE respectively. Line segment BD is a diameter of the circle with center F. What is the...
-
What is the relation between the square of the mean velocity and the mean of the velocity squared in the context of the Boltzmann distribution?
-
The distribution of the energy of the molecules is also of importance in the kinetics of chemical reactions. The fraction of molecules with energy in the range between \(E\) and \(E+d E\) is given...
-
The simple formula \(C_{p}=(5 / 2) R\) (molar units) is valid only for monatomic gases. A simple extension that has been suggested is \[C_{p}=\left(5+N_{\mathrm{r}} ight) \frac{1}{2} R\] where...

Study smarter with the SolutionInn App


