A gas evolved during a chemical reaction and has a volume of 612 mL at 34...
Fantastic news! We've Found the answer you've been seeking!
Question:
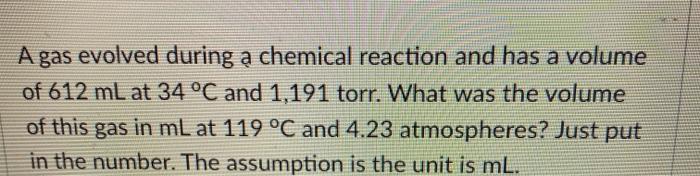

Transcribed Image Text:
A gas evolved during a chemical reaction and has a volume of 612 mL at 34 °C and 1,191 torr. What was the volume of this gas in mL at 119 °C and 4.23 atmospheres? Just put in the number. The assumption is the unit is mL. A sample of aluminum metal reacts completely with an excess of hydrochloric acid: 2 Al(s) + 6 HCl(aq) --> 2AlCl3(aq) + 3 H₂(g) The hydrogen gas produced is collected over water at 28.0 °C, so that there was a mixture of water vapor and hydrogen in the gas state. The water vapor was then removed with a drying agent so that the gas is pure hydrogen.The volume of the pure gas is 1,012 mL, and the total pressure in the gas collection vessel with just hydrogen, H₂ gas is 113.2 kpa (kilopascal). Calculate the mass of aluminum metal that reacted in grams. Just put in the number. The assumption is the unit is grams. 1 atm = 760 torr = 760 mmHg = 101,325 pa = 101.325 kpa 1.01325 barr = 14.696 p.s.i. = 29.921 inHg = (inHg and p.s.i. are approximations of the metric pressures and are subject to significant figures) A gas evolved during a chemical reaction and has a volume of 612 mL at 34 °C and 1,191 torr. What was the volume of this gas in mL at 119 °C and 4.23 atmospheres? Just put in the number. The assumption is the unit is mL. A sample of aluminum metal reacts completely with an excess of hydrochloric acid: 2 Al(s) + 6 HCl(aq) --> 2AlCl3(aq) + 3 H₂(g) The hydrogen gas produced is collected over water at 28.0 °C, so that there was a mixture of water vapor and hydrogen in the gas state. The water vapor was then removed with a drying agent so that the gas is pure hydrogen.The volume of the pure gas is 1,012 mL, and the total pressure in the gas collection vessel with just hydrogen, H₂ gas is 113.2 kpa (kilopascal). Calculate the mass of aluminum metal that reacted in grams. Just put in the number. The assumption is the unit is grams. 1 atm = 760 torr = 760 mmHg = 101,325 pa = 101.325 kpa 1.01325 barr = 14.696 p.s.i. = 29.921 inHg = (inHg and p.s.i. are approximations of the metric pressures and are subject to significant figures)
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A sample of zinc metal reacts completely with an excess of hydrochloric acid: Zn(s) + 2HCl(aq) ZnCl2(aq) + H2(g) The hydrogen gas produced is collected over water at 25.0C using an arrangement...
-
A piece of sodium metal reacts completely with water as follows: 2Na(s) + 2H2O(l) 2NaOH(aq) + H2(g) The hydrogen gas generated is collected over water at 25.0C. The volume of the gas is 246 mL...
-
An apple weighs 0.2 lbm and has a volume of 6 in3 in a refrigerator at 38 F. What is the apple density? List three intensive and two extensive properties for the apple?
-
The voltage held by a voltage regulator follows a normal random variable with a mean that equals 200 volts and a standard deviation that equals 5 volts. A regulator meets the specifications if the...
-
How can a company use change management to minimize the resistance and maximize the acceptance of changes in business and technology? Give several examples.
-
Midas and Company is the managing investment dealer for a major new underwriting. The price of the stock to the managing investment dealer is $15 per share. Other syndicate members may buy at $15.25....
-
Refer to Exercise 9. For the result that was statistically significant, the P-value was 0.01. What P-value would be needed to conclude at the = 0.05 level that the time spent studying differs...
-
A stock has a beta of 1.35 and an expected return of 16%. A risk-free asset currently earns 4.8%. 1. What is the expected return on a portfolio that is equally invested in the two assets? 2. If a...
-
Problem 14-3 (Algo) Straight-line and effective interest compared [LO14-2] On January 1, 2024, Reyes Recreational Products issued $100,000, 13%, four-year bonds. Interest is paid semiannually on June...
-
Annette Creighton opened Creighton Consulting. She rented a small office and paid a part-time worker to answer the telephone and make deliveries. Her chart of accounts is as follows: Creighton's...
-
I have seen an example of an emerging approach that has been successful in the commercial world. Competition from other businesses operating in the same industry was fierce for the company I worked...
-
Journalize the following transactions for Sandhill Company. (a) Purchased 6,200 units of raw materials on account for $17,750. The standard cost was $18,600. (b) Issued 5,990 units of raw materials...
-
determine the risk and return profile for your client, Jennifer, who is a moderate aggressive investor and willing and able to take risk, and wishes to allocate 75% of her funds on risky assets and...
-
Q4. You are given the following information about country A in period 0:r (interest rate paid on debt) = 5%; g (growth rate of GDP) = 9%; b (debt-to-GDP ratio) = 80%. (a) Calculate the primary...
-
How would partial bugeting look like when youre decudding whether to replace current seed with Td-away?
-
The Dashwoods have calculated their taxable income to be $88,000 for 2021, which includes $2,000 of long-term capital gains. Using the appropriate tax rate schedules, Dividends and Capital Gains Tax...
-
Jim Martin owns a men's clothing store in Philadelphia. He was recently offered the position of store manager at Anthony's, a large retail chain store in the area. Working at Anthony's, Jim would...
-
Akramin just graduated with a Master of Engineering in Manufacturing Engineering and landed a new job in Melaka with a starting salary of RM 4,000 per month. There are a number of things that he...
-
In the thorium decay series, thorium-232 loses a total of 6 particles and 4 particles in a 10-stage process. What is the final isotope produced?
-
Carbon has two stable isotopes, 126C and 136C, and fluorine has only one stable isotope, 199F. How many peaks would you observe in the mass spectrum of the positive ion of CF+4? Assume that the ion...
-
The pOH of a solution is 9.40. Calculate the hydrogen ion concentration of the solution.
-
A firm is being sued for \($550,000\) by an unhappy customer; the lawsuit is in its early stages, and the firm feels that it has a good case and is willing to defend itself. However, legal costs will...
-
Distinguish between the par value and the issue price of a bond. When are they the same? When are they different? Explain. If a \($5,000\) bond is sold for 101, how much cash is paid/received?
-
A company issues 10,000 coupons that allow a discount of \($1\) off the retail price of a \($14\) product. Will some liability be recorded? Explain.

Study smarter with the SolutionInn App


