Question: A gravitational pendulum makes n = 50 min cycles. What is the period, frequency and the average angular velocity of the pendulum? What is
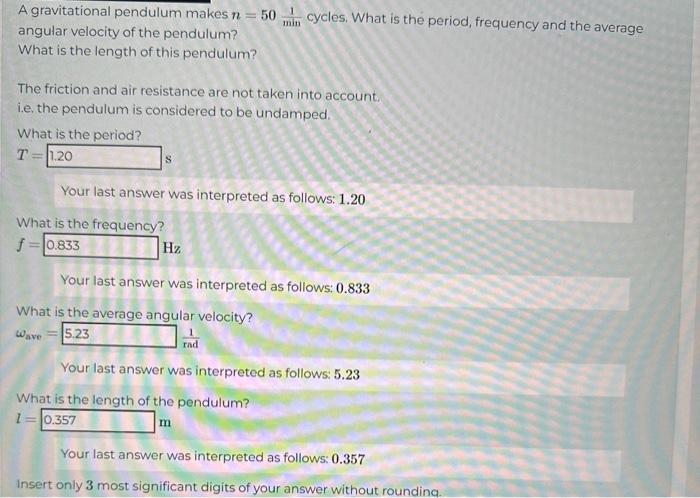
A gravitational pendulum makes n = 50 min cycles. What is the period, frequency and the average angular velocity of the pendulum? What is the length of this pendulum? The friction and air resistance are not taken into account. i.e. the pendulum is considered to be undamped. What is the period? T 1.20 S Your last answer was interpreted as follows: 1.20 What is the frequency? f = 0.833 Hz Your last answer was interpreted as follows: 0.833 What is the average angular velocity? Wave 5.23 Your last answer was interpreted as follows: 5.23 What is the length of the pendulum? 1=0.357 m Your last answer was interpreted as follows: 0.357 Insert only 3 most significant digits of your answer without rounding.
Step by Step Solution
There are 3 Steps involved in it
d In the given Question 50 cycles 1 min 60 sec i T... View full answer

Get step-by-step solutions from verified subject matter experts


