A LiBr/HO absorption chiller operates at the following temperatures: generator 95C; condenser 40C; evaporator 5C; and...
Fantastic news! We've Found the answer you've been seeking!
Question:
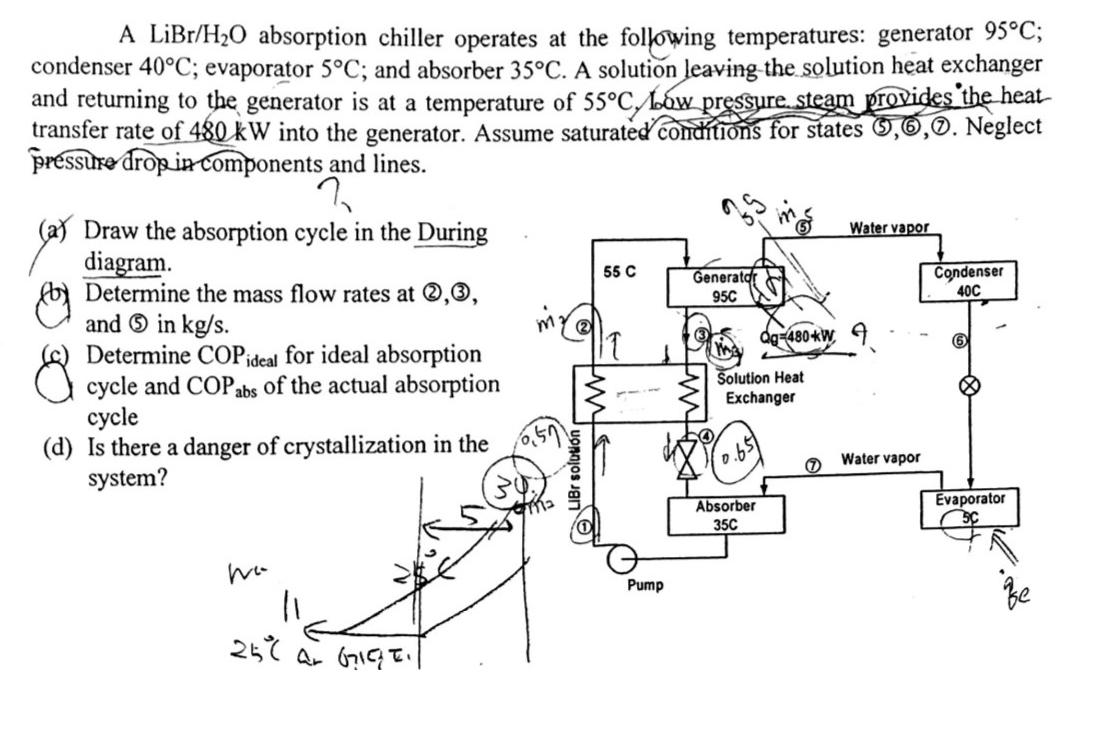
Transcribed Image Text:
A LiBr/H₂O absorption chiller operates at the following temperatures: generator 95°C; condenser 40°C; evaporator 5°C; and absorber 35°C. A solution leaving the solution heat exchanger and returning to the generator is at a temperature of 55°C Low pressure steam provides the heat transfer rate of 480 kW into the generator. Assume saturated conditions for states ,,Ⓒ. Neglect pressure drop in components and lines. % (a) Draw the absorption cycle in the During diagram. Determine the mass flow rates at 0,3, and in kg/s. Determine COP ideal for ideal absorption cycle and COP abs of the actual absorption cycle (d) Is there a danger of crystallization in the system? We 25 °C ar 6715E. Br solution 55 C 17 Pump Generator 95C Solution Heat Exchanger 0.65 Qg-480 kW Absorber 35C Water vapor Water vapor Condenser 40C 6 Evaporator Be A LiBr/H₂O absorption chiller operates at the following temperatures: generator 95°C; condenser 40°C; evaporator 5°C; and absorber 35°C. A solution leaving the solution heat exchanger and returning to the generator is at a temperature of 55°C Low pressure steam provides the heat transfer rate of 480 kW into the generator. Assume saturated conditions for states ,,Ⓒ. Neglect pressure drop in components and lines. % (a) Draw the absorption cycle in the During diagram. Determine the mass flow rates at 0,3, and in kg/s. Determine COP ideal for ideal absorption cycle and COP abs of the actual absorption cycle (d) Is there a danger of crystallization in the system? We 25 °C ar 6715E. Br solution 55 C 17 Pump Generator 95C Solution Heat Exchanger 0.65 Qg-480 kW Absorber 35C Water vapor Water vapor Condenser 40C 6 Evaporator Be
Expert Answer:
Answer rating: 100% (QA)
The LiBrHO absorption chiller operates at the following temperatures generator 95C condenser 40C evaporator 5C and absorber 35C Explanation A solution leaving the solution heat exchanger and returning ... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder, ? Ronald W. Rousseau, ? Lisa G. Bullard
Posted Date:
Students also viewed these organizational behavior questions
-
The LiBr water absorption cycle operates at the following temperature generator 110C, condenser 45C, evaporator 15C, and absorber 40c. The flow rate of solution delivered by the pump is 0.5 kg/s 1....
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Foreman Publishing Companys income for the most recent quarter was $500,000, and the average net book value of assets during the quarter was $1.5 million. If the company has a required rate of return...
-
On April 1, 2017, you prepaid three months of rent for a total of $18,000. Give your adjusting entry to record rent expense at April 30, 2017. Include the date of the entry and an explanation. Then,...
-
An article titled The Latest on Workplace Monitoring and Surveillance (American Management Association, November 17, 2014) referred to the 2007 Electronic Monitoring & Surveillance Survey. In a...
-
Four holes are drilled in a uniform circular disc at a radius of \(100 \mathrm{~mm}\) and angles of \(0^{\circ}, 60^{\circ}\), \(120^{\circ}\), and \(180^{\circ}\). The weight removed at holes 1 and...
-
(Multiple-step Statement with Retained Earnings) Presented below is information related to Ivan Calderon Corp. for the year 2004. Net sales $1,300,000 Write-off of inventory due to obsolescence $...
-
Describe the process termination sequence in an operating system. How does the OS ensure that resources are properly released, and what mechanisms are used to manage orphaned processes or zombie...
-
The beam shown in Figure P567 carries a repeated, reversed load of 400 N applied at section C. Compute the resulting design factor if the beam is made from SAE 1340 OQT 1300. Data in Figure P567. 12...
-
1 What is the utopian value of Jims venture? Provide all details of your calculations. 2 What is the P/E multiple utopian value of Jims venture? Provide all details of your calculations. 3 What is...
-
The couple currently has $7,350 in their savings account. Each month, the couple will take money out of their savings account or deposit money into their savings account. calculate the end-of-month...
-
For each of the following systems, determine whether they are static or dynamic linear or non-linear ii) v) time-invariant or time-varying causal or non-causal Stable or unstable. Hints: a. if you...
-
Cash dividends of $33,500 were paid during the year. Retained earnings as of September 1, 20Y5, were $238,000. Prepare a balance sheet as of August 31, 20Y6. When entering assets, enter them in order...
-
Given the circuit shown in figure 1 with component values R = 10 kn, C = 1 F and L = 22mH. Voltage sources V = 3 V (DC) and V = 1.732 +j V (AC) with frequency f1 [Hz] which is not yet defined. Assume...
-
You are given a list of integers that are not sorted. Your job is to sort the list in a nondecreasing order. You should implement four different sorting algorithms. algorithm 1: choose one of the...
-
A subway train travels between two stations with the acceleration graph shown. Determine the time interval, A? such that the train brakes to a stop with a deceleration of 2 m/s, and finds the...
-
The Adjusted Trial Balance columns of a 10-column work sheet for Webber Co. follow. Complete the work sheet by extending the account balances into the appropriate financial statement columns and by...
-
Under the FutureGen 2.0 project (www.futuregenalliance.org/) sponsored by the U.S. Department of Energy, a novel process is used to convert coal into electricity with minimal greenhouse gas (CO2)...
-
Lewis describes the hazards of breathing air containing appreciable amounts of an asphyxiant (a gas that has no specific toxicity but, when inhaled, excludes oxygen from the lungs). When the mole...
-
Strawberries contain about 15 wt% solids and 85 wt% water. To make strawberry jam, crushed strawberries and sugar are mixed in a 45:55 mass ratio, and the mixture is heated to evaporate water until...
-
Find the steady-state response of the system shown in Fig. 3.55 for the following data: \(k_{1}=1000 \mathrm{~N} / \mathrm{m}, k_{2}=500 \mathrm{~N} / \mathrm{m}, c=500 \mathrm{~N}-\mathrm{s} /...
-
A uniform slender bar of mass \(m\) may be supported in one of two ways as shown in Fig. 3.56. Determine the arrangement that results in a reduced steady-state response of the bar under a harmonic...
-
By denoting the amplitudes of velocity and acceleration of the mass of a viscously damped system subjected to a harmonic force, as \(\dot{X}\) and \(\ddot{X}\), respectively, find expressions for the...

Study smarter with the SolutionInn App


