?? A medical technician is working with the four samples of radionuclides listed in the table below.
Fantastic news! We've Found the answer you've been seeking!
Question:

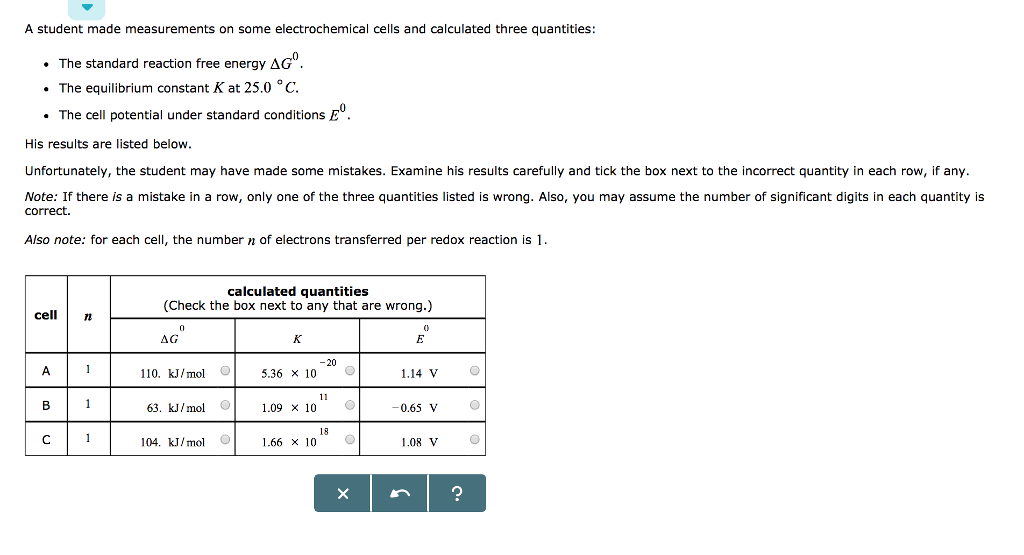
Transcribed Image Text:
A medical technician is working with the four samples of radionuclides listed in the table below. Initially, each sample contains 5.00 μmol of the radionuclide. First, order the samples by decreasing initial radioactivity. Then calculate how long will take for the amount of radionuclide in each sample to decrease to 1/32 of the initial amount. sample A B с D symbol 152 63 Eu 99 42 Mo 82 37 Rb radionuclide 192 77 Ir half-life 13 years. 66.0 hours 1 minute 74.0 days initial radioactivity (choose one) (choose one) + (choose one) # (choose one) time for amount of radionuclide to decrease to 1/32 of Initial amount X years hours minutes days ? A student made measurements on some electrochemical cells and calculated three quantities: • The standard reaction free energy AG⁰. The equilibrium constant K at 25.0 °C. • The cell potential under standard conditions º His results are listed below. Unfortunately, the student may have made some mistakes. Examine his results carefully and tick the box next to the incorrect quantity in each row, if any. Note: If there is a mistake in a row, only one of the three quantities listed is wrong. Also, you may assume the number of significant digits in each quantity is correct. Also note: for each cell, the number of electrons transferred per redox reaction is 1. cell 72 A B с 1 1 1 calculated quantities (Check the box next to any that are wrong.) AG 0 110. kJ/mol O 63. kJ/mol 104. kJ/mol K -20 5.36 x 10 C 1.09 x 10 11 18 1.66 x 10. O O X E 1.14 V -0.65 V 1.08 V ? O O O A medical technician is working with the four samples of radionuclides listed in the table below. Initially, each sample contains 5.00 μmol of the radionuclide. First, order the samples by decreasing initial radioactivity. Then calculate how long will take for the amount of radionuclide in each sample to decrease to 1/32 of the initial amount. sample A B с D symbol 152 63 Eu 99 42 Mo 82 37 Rb radionuclide 192 77 Ir half-life 13 years. 66.0 hours 1 minute 74.0 days initial radioactivity (choose one) (choose one) + (choose one) # (choose one) time for amount of radionuclide to decrease to 1/32 of Initial amount X years hours minutes days ? A student made measurements on some electrochemical cells and calculated three quantities: • The standard reaction free energy AG⁰. The equilibrium constant K at 25.0 °C. • The cell potential under standard conditions º His results are listed below. Unfortunately, the student may have made some mistakes. Examine his results carefully and tick the box next to the incorrect quantity in each row, if any. Note: If there is a mistake in a row, only one of the three quantities listed is wrong. Also, you may assume the number of significant digits in each quantity is correct. Also note: for each cell, the number of electrons transferred per redox reaction is 1. cell 72 A B с 1 1 1 calculated quantities (Check the box next to any that are wrong.) AG 0 110. kJ/mol O 63. kJ/mol 104. kJ/mol K -20 5.36 x 10 C 1.09 x 10 11 18 1.66 x 10. O O X E 1.14 V -0.65 V 1.08 V ? O O O
Expert Answer:
Answer rating: 100% (QA)
Lower the half life higher will be initial radioactivity so initial radio activity Samp... View the full answer

Posted Date:
Students also viewed these chemistry questions
-
The carbon steels listed in the table below were soaked at 1000 C for 1 hour to form austenite and were cooled slowly, under equilibrium conditions to room temperature. Refer to the Fe-Fe3C phase...
-
Lets figure out how long it will take for the average Indian to be as wealthy as the average Western European is today. All numbers are adjusting for inflation, so were measuring output in piles of...
-
Mia Salto wishes to determine how long it will take to repay a loan with initial proceeds of $14,000 where annual end-of-year installment payments of $2,450 are required. a. If Mia can borrow at a...
-
Write the C++ code for a function that receives an integer, a double number, and the address of a double variable from the calling statement. The function should multiply the integer by the double...
-
What does "property" include for purposes of 351?
-
Tenney Construction Company is considering selling excess machinery with a book value of $210,000 (original cost of $320,000 less accumulated depreciation of $110,000) for $180,000 less a 5%...
-
Two objects A and B having inertias \(m_{a}=m\) and \(m_{b}=3 m\) are moving with velocities \(v_{a}=v\) and \(v_{b}=3 v\). Find the velocities of the two objects in the zero-momentum frame of...
-
Portman Corporation has retained earnings of $675,000 at January 1, 2010 Net income during 2010 was $1,400,000, and cash dividends declared and paid during 2010 totaled $75,000. Prepare a retained...
-
_____ redirects a user from a legitimate website to a malicious website by changing hosts files on a DNS server. A. Exploitation framework B. ARP poisoning C. DDoS DNS attack D. DNS...
-
A test of tennis serve accuracy resulted in the following data: Note: Higher scores indicate better performance. X 15 12 12 10 8 7 5 4 4 1. What is the percentile rank of a person who scored (a) 7;...
-
Heap sort program to sort array in descending order using MinHeap.
-
Ozark Cabin Resorts rents vacation cabins to tourists. Customers pay at the time they book their reservations. In 2020, Ozark Cabin Resorts had a beginning balance of $8,300 in its Unearned Service...
-
Brighton Company has $920 of supplies reported on its unadjusted trial balance as of August 30. At the end of August, Brighton Company had $450 worth of supplies remaining. What is the entry to...
-
Explain the fundamental differences between science and pseudo-science. also what is the ultimate goal of it? , what is the role of observable reality?, how's it willing to change options as new...
-
The POL Consulting Company has the following professional labor costs for the most recent year: Budget Actual Total Clients 75 70 Total Professional Labor Hours 4,500 4,340 Total Professional Labor...
-
He was given the option to live at the lodge if he would also look after the night auditing (the value of his reviews is $500 per month) responsibilities. The value of the motel unit on a monthly...
-
Journal Entry 2 (minimum of 250 words) Reflect on an important event or incident that you observed or were involved in durin your Fieldwork Placement. For example: What was the event? How did it...
-
Grace is training to be an airplane pilot and must complete five days of flying training in October with at least one day of rest between trainings. How many ways can Grace schedule her flying...
-
Frieda Wannamaker is a taxable investor who is currently in the 28% income-tax bracket. She is considering purchasing a tax-exempt bond with a yield of 3.75%. The taxable equivalent yield on this...
-
A rationale for the use of the price-to-sales (P/S) approach is: a. Sales are more volatile than earnings. b. P/S ratios assess cost structures accurately. c. Revenues are less subject to accounting...
-
Technical analysis looks at the demand and supply for securities based on trading volumes and price studies. Charting is a common method used to identify and project price trends in a security. A...

Study smarter with the SolutionInn App

