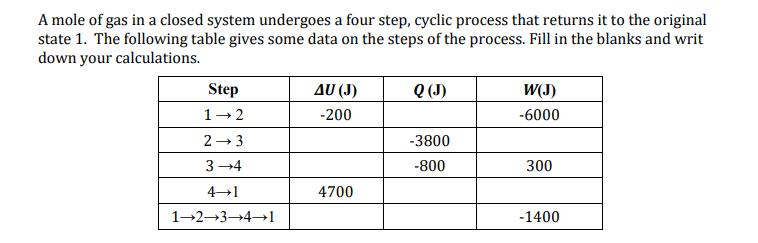
A mole of gas in a closed system undergoes a four step, cyclic process that returns...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A mole of gas in a closed system undergoes a four step, cyclic process that returns it to the original state 1. The following table gives some data on the steps of the process. Fill in the blanks and writ down your calculations. Step 1-2 2-3 3-4 4-1 1-2-3-4-1 AU (J) -200 4700 Q (J) -3800 -800 W(J) -6000 300 -1400 A mole of gas in a closed system undergoes a four step, cyclic process that returns it to the original state 1. The following table gives some data on the steps of the process. Fill in the blanks and writ down your calculations. Step 1-2 2-3 3-4 4-1 1-2-3-4-1 AU (J) -200 4700 Q (J) -3800 -800 W(J) -6000 300 -1400
Expert Answer:
Related Book For 

Introduction to Chemical Engineering Thermodynamics
ISBN: 978-0071247085
7th edition
Authors: J. M. Smith, H. C. Van Ness, M. M. Abbott
Posted Date:
Students also viewed these mechanical engineering questions
-
One mole of gas in a closed system undergoes a four-step thermodynamic cycle. Use the data given in the following table to determine numerical values fur the missing quantities, i.e., "fill in the...
-
The table gives some data on the demand for food. Calculate the income elasticity of demand for food when the price of food is $50 a unit. When the price of food is $50 a unit, the income elasticity...
-
A mole of gas at 0oC and 760 mmHg occupies 22.41 L. What is the volume at 20oC and 760 mmHg?
-
Formulate a plausible mechanism for the following reaction. The product is a precursor of mediquox (shown in the margin), an agent used to treat respiratory infections in chickens (no, we are not...
-
Anheuser-Busch InBev is attempting to reduce its water usage. How could a company manager use a process cost summary to determine if the program to reduce water usage is successful?
-
The instrument package shown in Fig. 5.18 weighs 258 N. Calculate the tension in the cable if the package is completely submerged in seawater having a specific weight of 10.05 kN/m 3 . Water surface...
-
Who can conduct cost audit under the Companies Act, 2013?
-
When (R)-(+)-2-phenyl-2-butanol is allowed to stand in methanol containing a few drops of sulfuric acid; racemic 2-methoxy-2-phenylbutane is formed. Suggest a reasonable mechanism for this reaction.
-
What are the two basic timing strategies? What is the intent of each? If nontax factors are irrelevant, what is the best course of action in timing strategy when you expect tax rate to increase in...
-
Pathfinder College is a small liberal arts college that wants to improve its admissions process. In particular, too many of its incoming freshmen have failed to graduate for a variety of reasons,...
-
What reagent would be used to carry out this reaction? OH O chromic acid peracetic acid O hydrogen peroxide O osmium tetroxide O periodic acid Question 9 What reagent would be used to carry out this...
-
The following information concerns production in the Baking Department for August. All direct materials are placed in process at the beginning of production. ACCOUNT Work in Process-Baking Department...
-
Entries for issuing and calling bonds; gain Mia Breen Company produces and sells wind-energy-driven engines. To finance its operations, Mia Breen issued $1,854,000 of 10-year, 10% callable bonds on...
-
1. Assuming McCullough continues to use only one predetermined overhead rate, calculate: a. The predetermined overhead rate. b. The total cost, including direct materials, direct labor, and applied...
-
ET Mag Wheels of Benicia, California had the following transactions during the month of July 2 0 2 3 : Purchased raw materials on account, $ 9 1 , 6 0 5 . Raw Materials of $ 7 6 , 0 5 0 were...
-
Lok Company reports net sales of $5,096,000 for Year 2 and $8,788,000 for Year 3. End-of-year balances for total assets are Year 1, $1,503,000; Year 2, $1,792,000; and Year 3, $1,926,000. (1) Compute...
-
On 1 st December 2018, Night Limited acquired all the assets (except cash and cash equivalent) and current liabilities of Day Limited. The fair values of Day Limited's assets and liabilities at this...
-
What is taxable income, and what is the formula for determining taxable income?
-
Estimate (H((298 for one of the following compounds as a liquid at 25(C. (a) Acetylene, (b) 13-Butadiene, (c) Ethylbenzene, (d) n-Hexane, (e) Styrene.
-
The contents of the freezer in a home refrigerator are maintained at - 20C. The kitchen temperature is 20C. If heal leaks amount to 125.000 U per day and if electricity costs $0.08/k Whr, estimate...
-
If the heat of combustion of urea, (NH2)2CO(s), at 25(C is 631 660 J mol-1 when the products are CO2(g), H2O(l), and N2(g), what is (H((298 for urea?
-
Calculate the evolution operator for the interaction picture in the case in exercise 4. Data From Exercise 4:- Consider a harmonic oscillator perturbed by \(H_{1}=\lambda\left(a+a^{\dagger}...
-
Write down the time evolution equation for Heisenberg operators, calculating explicitly the evolution Hamiltonian, in the case of a free particle, and then calculate explicit expressions for...
-
Write down the time evolution operator and the differential equation that it satisfies for a onedimensional particle in a potential \(V(x)\).

Study smarter with the SolutionInn App



