Question: A particle in a doop square well patential extende from x=0 to x = L as a walle free Y(x)= // = [19> ++212]
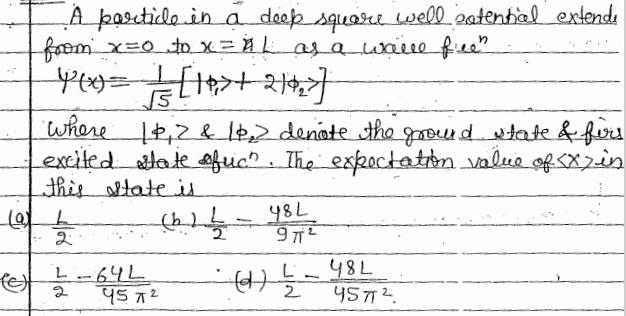
A particle in a doop square well patential extende from x=0 to x = L as a walle free" Y(x)= // = [19> ++212] where 182 & 1p, denote the ground state & firs excited state fuc". The expectation value of SX) in this state is (b) b = (9) L 2 L 2 -64L - 45 72 48L 972 - -(1) // - 2 48L 457
Step by Step Solution
★★★★★
3.49 Rating (159 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

The detailed ... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


