Pressure sensor A piston with diameter 1-cm and a frictionless plunger is filled with a diatomic...
Fantastic news! We've Found the answer you've been seeking!
Question:
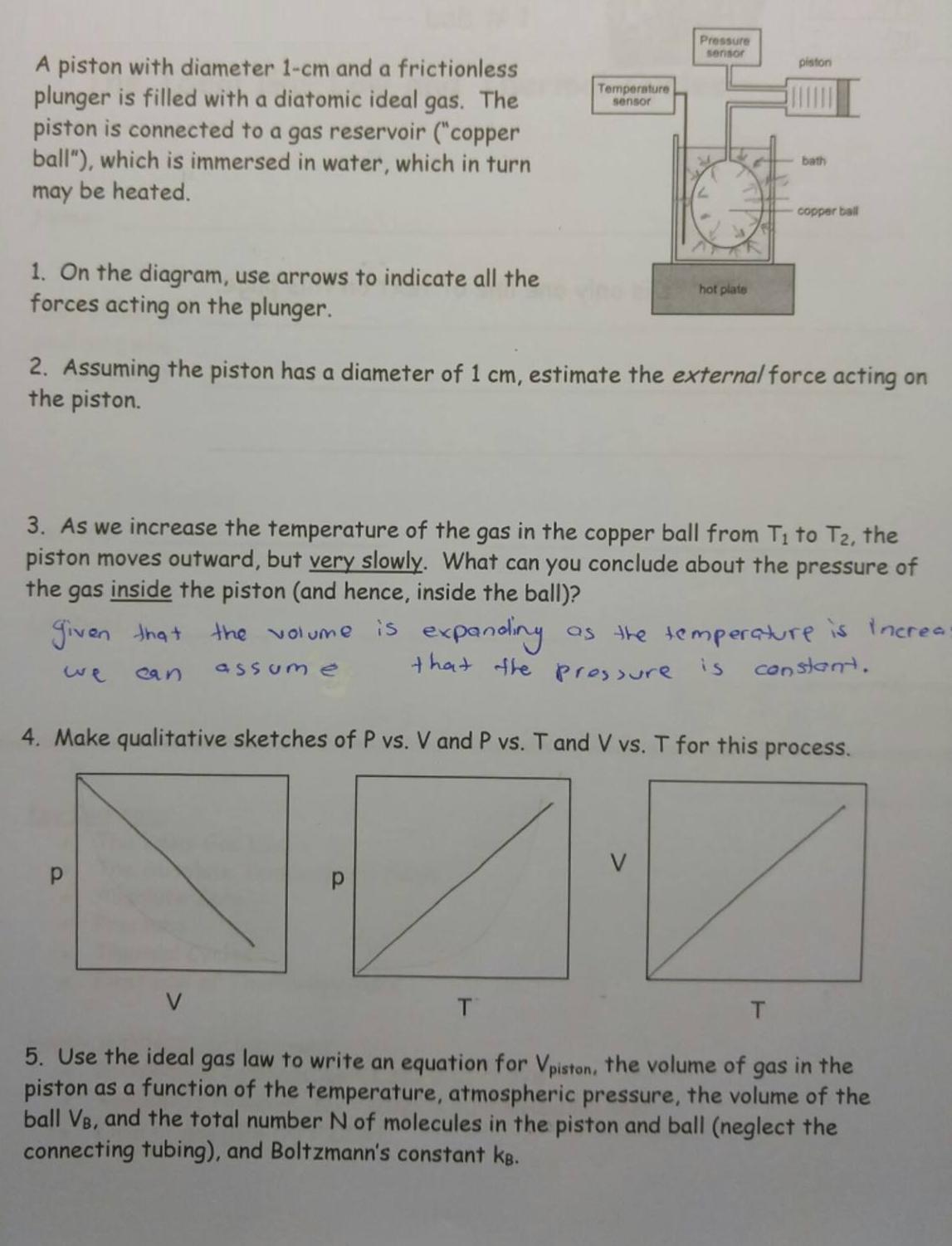
Transcribed Image Text:
Pressure sensor A piston with diameter 1-cm and a frictionless plunger is filled with a diatomic ideal gas. The piston is connected to a gas reservoir ("copper ball"), which is immersed in water, which in turn may be heated. piston Temperature sensor bath copper ball 1. On the diagram, use arrows to indicate all the forces acting on the plunger. hot plate 2. Assuming the piston has a diameter of 1 cm, estimate the external force acting on the piston. 3. As we increase the temperature of the gas in the copper ball from T1 to T2, the piston moves outward, but very slowly. What can you conclude about the pressure of the gas inside the piston (and hence, inside the ball)? is expanding as the temperature is increa that the prossure is constant. Jiven that the volume we ean assum e 4. Make qualitative sketches of P vs. V and P vs. T and V vs. T for this process. T. T. 5. Use the ideal gas law to write an equation for Vpiston, the volume of gas in the piston as a function of the temperature, atmospheric pressure, the volume of the ball VB, and the total number N of molecules in the piston and ball (neglect the connecting tubing), and Boltzmann's constant kB. Pressure sensor A piston with diameter 1-cm and a frictionless plunger is filled with a diatomic ideal gas. The piston is connected to a gas reservoir ("copper ball"), which is immersed in water, which in turn may be heated. piston Temperature sensor bath copper ball 1. On the diagram, use arrows to indicate all the forces acting on the plunger. hot plate 2. Assuming the piston has a diameter of 1 cm, estimate the external force acting on the piston. 3. As we increase the temperature of the gas in the copper ball from T1 to T2, the piston moves outward, but very slowly. What can you conclude about the pressure of the gas inside the piston (and hence, inside the ball)? is expanding as the temperature is increa that the prossure is constant. Jiven that the volume we ean assum e 4. Make qualitative sketches of P vs. V and P vs. T and V vs. T for this process. T. T. 5. Use the ideal gas law to write an equation for Vpiston, the volume of gas in the piston as a function of the temperature, atmospheric pressure, the volume of the ball VB, and the total number N of molecules in the piston and ball (neglect the connecting tubing), and Boltzmann's constant kB.
Expert Answer:
Answer rating: 100% (QA)
a 2 External force acting on the the piston F ext is due to atmospheric pressureP atm so F ext F atm ... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these accounting questions
-
What can you conclude about the price elasticity of demand in each of the following statements? a. "The pizza delivery business in this town is very competitive. I'd lose half my customers if I...
-
What can you conclude about the relative risk of investing in the United States versus Japan from Figure 7.4?
-
What can you conclude about the ratio of fugacity to pressure for N 2 ,H 2 , and NH 3 at 500 bar using the data in Figure 7.10? Figure 7.10 1.5 H2, 1.4 N2 1.3 1.2 1.1 1.0 100 200 300 400 500 600 700...
-
A clinical study is established to determine if the results of a screening stress test can be used as a predictor of the presence of heart disease. The study enrolls 100 participants who undergo a...
-
The incomes of all families in a particular suburb can be represented by a continuous random variable. It is known that the median income for all families in this suburb is $60,000 and that 40% of...
-
A 2.4-L gasoline fuelled internal combustion engine produces a maximum power of 200 hp when the fuel is burned stoichiometrically with air. The thermal efficiency of the engine at this maximum power...
-
Use the $\mathrm{FrF} 2():: \mathrm{FrF} 2()$ function to generate the design matrix in the standard order for a $2^{3}$ design with blocks generated using the three-way interaction.
-
During December, Far West Services makes a $3,200 credit sale. The state sales tax rate is 6% and the local sales tax rate is 2.5%. Record sales and sales tax payable.
-
KJSC , Inc. - Using the following information, create journal entries and t - accounts ON A SEPARATE PIECE OF PAPER. PLEASE NOTE - these facts will only be presented in this question. You will need...
-
Arcadia, Inc., acquired 100 percent of the voting shares of Bruno Company on January 1, 2023. In exchange, Arcadia paid $198,000 in cash and issued 100,000 shares of its own $1 par value common...
-
In the production mix problem, the meaning for the value of a dual variable is the marginal increase of the profit for the furniture company from one unit increase in the corresponding resource....
-
On December 31, 2018, Diaz Corp. is in financial difficulty and cannot pay a $900,000 note with $90,000 accrued interest payable to Cameron Ltd., which is now due. Cameron agrees to accept from Diaz...
-
bill and dan opened up HEB Inc. on September 1, 2013. During the first month of operations the following transactions occurred. Date Transaction 9-1-13 Common Stockholders invested $40,000 cash in...
-
As a General Manager, you are tasked with leading a company through a significant transition period that involves restructuring, cultural change, and technological upgrades. Describe the...
-
Problem 3: An accountant, Keepallyer Munny, had been losing business to a competitor down the street because of TV ads put out that said the lines were too long and the turnaround time was to slow at...
-
1 - Consider a typical laptop. Offer examples of items related to the four quadrants of the Kralic Matrix. Explain your choices. Explain how your categorization differentiates to your selection of...
-
Luckie Company provides complementary breakfast to all employees. This is considered a/an incentive O health care cost O benefit plan O perquisite 1 pt
-
CdF2 (s) Cd+ (aq) + 2 F- (aq) 1. A saturated solution of CdF2 is prepared. The equilibrium in the solution is represented above. In the solution [Cd+] eq = 0.0585 M and [F-] eq = 0.117 M. a....
-
Consider a TV set that consumes 120 W of electric power when it is on and is kept on for an average of 6 hours per day. For a unit electricity cost of 12 cents per kWh, determine the cost of...
-
An ideal gas under goes a constant pressure (isobaric) process in a closed system. The heat transfer and work are, respectively (a) 0, cvT (b) cvT, 0 (c) cpT, RT (d) R ln(T2/T1), R ln(T2/T1)
-
Reconsider Prob. 6-45. Using EES (or other) software, determine the power input required by the air conditioner to cool the house as a function for airconditioner EER ratings in the range 5 to 15....
-
Pick any three of the sources of prospects discussed in the chapter and pick a product or service you like. Develop several ideas for how you would use each source to locate leads for the product or...
-
Why do you think a salesperson might experience call reluctance? How can it be overcome?
-
List three or four criteria you could use to qualify a lead as a likely prospect. How would you find out if the lead meets these criteria?

Study smarter with the SolutionInn App


