A pistoncylinder device (with a piston diameter of 3 mm) is used to fill a balloon with
Question:
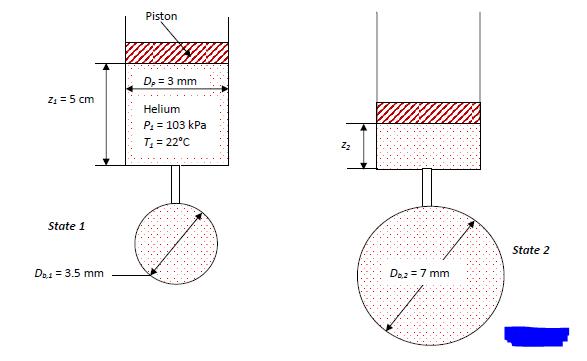
A piston‐cylinder device (with a piston diameter of 3 mm) is used to fill a balloon with helium as shown in the figure below. The pressure and temperature at the initial state (state 1) are 103 kPa and 22°C, respectively. The balloon stretches as it fills and so the pressure in the balloon increases. The pressure‐volume relationship within the balloon is given by: P = Patm + KbVb, where Patm is the atmospheric pressure (101.325 kPa) and Kb is a constant. The final diameter of the balloon is 7 mm.
The temperature of the helium in the cylinder is maintained at Tin = T1 throughout the process (i.e., all of the helium entering the balloon is at Tin) and the final temperature of helium in the balloon is T2 = 30°C. Model the helium as an ideal gas with R = 2.076 kJ/kg‐K. Assume that the cylinder and the balloon are both leak‐proof and that the pressure in the cylinder and the pressure in the balloon are always the same. Ignore the volume of the tube connecting the balloon to the cylinder.
a. What is the work done by the helium on the balloon during the filling process?
b. What is the mass of helium that was added to the balloon in order to inflate it?
c. What is the final position of the piston (z2)?
d. What is the heat transfer from the balloon to the surroundings (assume the helium in the balloon is an open system for this purpose)?

College Algebra Graphs and Models
ISBN: 978-0321845405
5th edition
Authors: Marvin L. Bittinger, Judith A. Beecher, David J. Ellenbogen, Judith A. Penna




