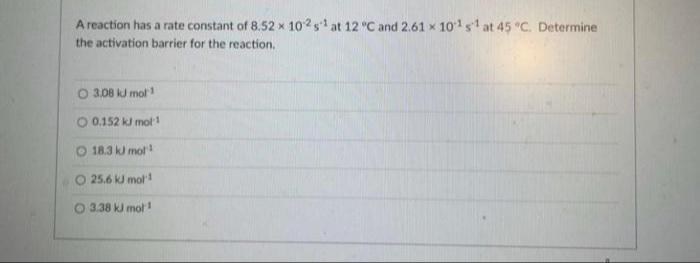
A reaction has a rate constant of 8.52 x 102s at 12 C and 2.61 x...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A reaction has a rate constant of 8.52 x 102s at 12 °C and 2.61 x 101s at 45 "C. Determine the activation barrier for the reaction. O 3.08 kJ mol 0.152 ki mot! O 18.3 k mol 25.6 kJ mol 3.38 kJ mot! A reaction has a rate constant of 8.52 x 102s at 12 °C and 2.61 x 101s at 45 "C. Determine the activation barrier for the reaction. O 3.08 kJ mol 0.152 ki mot! O 18.3 k mol 25.6 kJ mol 3.38 kJ mot!
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A second-order reaction has a rate constant of 8.7 104/(Ms) at 30oC. At 40oC, the rate constant is 1.8 103/(Ms). What are the activation energy and frequency factor for this reaction? Predict the...
-
(a) A certain first-order reaction has a rate constant of 2.75 10-2 s-1 at 20 oC. What is the value of k at 60 oC if? (b) Another first-order reaction also has a rate constant of 2.75 10-2 s-1 at...
-
Consider the diode bridge rectifier shown below with the voltage regulating capacitor in parallel with the load resistor. 60Hz 120V 10:1 C-470F IL R=1kQ a) For the input signal, sketch waveforms of...
-
What will the following code display? numbers = [1, 2, 3, 4, 5] numbers [2] = 99 print (numbers)
-
The farmer in Problem 3-25 has an opportunity to take over the neighboring 80-acre farm. If he acquires this farm, he will be able to increase the amounts of time available to 1,600 hours for...
-
Dan Luther Company has the following balances in selected accounts on December 31, 2014. Accounts Receivable .................. $ 0 Accumulated DepreciationEquipment .......... 0 Equipment...
-
In July 2017, Latrice Merritt entered a residential lease with Doran 610 Apartments, LLC. Under the terms of the lease agreement, Merritt was prohibited from installing a private security system in...
-
Presented below is information related to equipment owned by Suarez Company at December 31, 2007. Cost ............... $9,000,000 Accumulated depreciation to date ... 1,000,000 Expected future net...
-
2 1. Let I = S 1 + exc - - ex -dx. 1+ ex 0
-
Mike's car has an actual cash value of $10,000. While driving to class, at GBC, Mike was involved in an accident and is 25% responsible. The car is a total loss. If Mike has optional loss coverage,...
-
Find Area of the circle - Take input from the user for radius - print the value to the screen - use comments to explain what you are doing. 2) Put a program that calculates the total amount of a meal...
-
A woman was on her deathbed. There was one drug that the doctors thought might save her. It was a form of radium that a druggist in the same town had recently discovered. The drug was expensive to...
-
Dwight will incrased his 401(k) contribution to a 8% with a company matchof 50% up to 6% of the company's anuual salary. 50,000, the employer's match would not exceed 3,000..(50,000 x 6%). how much...
-
"Civil Law versus Common Law" Please respond to the following: Predict the long-term use of both civil and common law and explain whether you believe both have a foundation strong enough to last in...
-
A stock had returns of 12%, 2%, 9%, 16%, and -7% for the past five years. The average return on corporate bonds and government of Canada treasury bills over the past five years was 5% and 2.5%...
-
As compared to investment-grade bonds, "junk bonds" are characterized by Question 33 options: higher default risk lower coupon rate all answers are correct lower yield to maturity
-
As most plant cells lack lysosomes, what organelle in plant cells performs the functions of recycling and degradation of molecules and old organelles? O chloroplast O central vacuole O starch...
-
Prairie Outfitters, Inc., a retailer, accepts paymnent through credit cards. During August, credit card sales amounted to $12,000. The processor charges a 3% fee. Assuming that the credit card...
-
For each of the following equations, give the expression for the equilibrium constant Kc: a. NH4CIs)NH3(g) + HCI(g) b. 2N2(g) CO2(g)2N20g) C(s) c. 2NaHCO3(s)Na CO3(s) + H20(g) CO2(g) d. Fe + 30H...
-
What must be the concentration of sulfate ion in order to precipitate calcium sulfate, CaSO4, from a solution that is 0.0030 M Ca2+?
-
Hydrogen sulfide, H2S, is produced during decomposition of organic matter. When 0.5500 mol H2S burns to produce SO2(g) and H2O(l), 309.1 kJ of heat is released. What is this heat in kilocalories?
-
Figure 7.33 shows a thin-walled glass of milk, which is taken out of the refrigerator at a uniform temperature of \(3^{\circ} \mathrm{C}\) and is placed in a large pan filled with hot water at...
-
The junction of a thermocouple can be approximated as a sphere with a diameter of \(1 \mathrm{~mm}\). As shown in Figure 7.32, the thermocouple is used to measure the temperature of a gas stream. For...
-
Figure 7.46 represents the temperature dynamics of two adjacent objects, where the thermal capacitances of the objects are \(C_{1}\) and \(C_{2}\), respectively. Assume that the temperatures of both...

Study smarter with the SolutionInn App



