1. A sample of a concentrated sulfuric acid solution with a density of 1.84 g/cm contains...
Fantastic news! We've Found the answer you've been seeking!
Question:
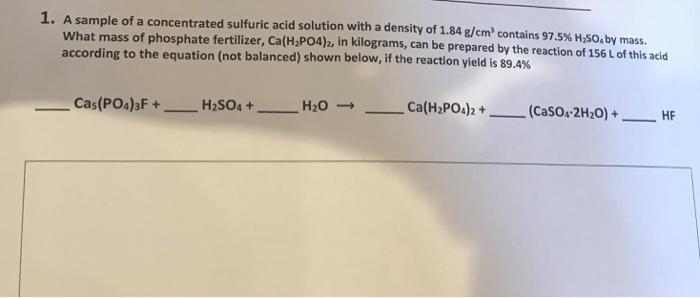
Transcribed Image Text:
1. A sample of a concentrated sulfuric acid solution with a density of 1.84 g/cm³ contains 97.5% H₂SO, by mass. What mass of phosphate fertilizer, Ca(H₂PO4)2, in kilograms, can be prepared by the reaction of 156 L of this acid according to the equation (not balanced) shown below, if the reaction yield is 89.4% Cas(PO4)3F+ H₂SO4 + — H₂O -> Ca(H₂PO4)2 + (CaSO4-2H₂O) + HF 1. A sample of a concentrated sulfuric acid solution with a density of 1.84 g/cm³ contains 97.5% H₂SO, by mass. What mass of phosphate fertilizer, Ca(H₂PO4)2, in kilograms, can be prepared by the reaction of 156 L of this acid according to the equation (not balanced) shown below, if the reaction yield is 89.4% Cas(PO4)3F+ H₂SO4 + — H₂O -> Ca(H₂PO4)2 + (CaSO4-2H₂O) + HF
Expert Answer:
Answer rating: 100% (QA)
2 CasPoub F HSO4 14 HO 36 12804 765042420 First balance Ply then Ca then s... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
A ketone can be prepared from the reaction of a nitrile with a Grignard reagent. Describe the intermediate that is formed in this reaction, and explain how it can be converted to a ketone.
-
Phosphoric acid, H3PO4, can be prepared by the reaction of phosphorus(V) oxide, P4O10, with water. What is H for the reaction involving 1 mol of P4O10? P4O10(s) + 6H2O(l ) 4H3PO4(aq) P,O10(s) +...
-
Gaseous iodine pentafluoride, IF5, can be prepared by the reaction of solid iodine and gaseous fluorine: I2 (s) + 5F2 (g) 2IF5 (g) A 5.00-L flask containing 10.0 g I2 is charged with 10.0 g F2, and...
-
Alert Security Services Co. offers security services to business clients. The trial balance for Alert Security Services Co. has been prepared on the following end-of-period spreadsheet for the year...
-
Compare the liquidity of an investment in raw land with that of an investment in common stock. Be specific as to why and how the liquidity differs.
-
Discuss four important risk and tax-related considerations that are relevant to Omos portfolio. With most of Omos personal net worth tied up in his software company, he is asset rich but cash poor....
-
Fun-Tastic Shows, Inc., is a company that hosts carnivals and similar events. Susan Swartwood, Crystal Groth, and a minor (named in the case as M.G.S.) attended Fun-Tastics Rhododendron Festival in...
-
Mark Price Company uses the gross profit method to estimate inventory for monthly reporting purposes. Presented below is information for the month of May. Inventory, May 1...
-
8. Two dice are rolled. Let A represent rolling a sum greater than 7. Let B represent rolling a sum that is a multiple of 3. Determine n(AB). A. 5 B. 8 C. 12 D. 15 Long Answer: Show all work for the...
-
Ray Flagg took out a 60-month fixed installment loan of $12,000 to open a new pet store. He paid no money down and began making monthly payments of $232. Ray's business does better than expected and...
-
Read the case The Decision Making Process and describe: The main phases or activities of the decision making process, including for example: the background leading up to the problem situation;...
-
Define cointegration.
-
Choose an organisation with which you are familiar and, using Guests evaluation of the four possible approaches to the management of the employment relationship, describe the current employment...
-
What is the purpose of moving average and exponential smoothing?
-
Describe the conditions under which MRP is most appropriate.
-
Fox (1966) clearly believed that the pluralistic perspective on employee relations was the most valid and realistic way to manage the employment relationship. Given the emergence of the HRM models of...
-
The rotor in the figure is supported by L and R at a constant speed of 10000 rpm around the z-axis. When it is rotated, the forces (FL and FR) coming from the bearings to the rotor are in the xy...
-
Sandcastles, Inc.s management has recently been looking at a proposal to purchase a new brick molding machine. With the new machine, the company would not have to buy bricks. The estimated useful...
-
If an average O3 molecule "lives" only 100-200 seconds in the stratosphere before undergoing dissociation, how can O3 offer any protection from ultraviolet radiation?
-
(a) Use data in Appendix C to estimate the boiling point of benzene, C6H6(l). (b) Use a reference source, such as the CRC Handbook of Chemistry and Physics, to find the experimental boiling point of...
-
Each of the following elements is capable of forming an ion in chemical reactions. By referring to the periodic table, predict the charge of the most stable ion of each: (a) Mg, (b) Al, (c) K, (d) S,...
-
A second-order dynamic system is modeled as \[9 \ddot{x}+6 \dot{x}+\frac{10}{9} x=14 \delta(t), \quad x(0)=0, \quad \dot{x}(0)=-\frac{1}{4}\] a. Find the response \(x(t)\) in closed form. b. Plot the...
-
Consider a first-order system with time constant \(\tau\) and zero initial condition. Find the system's unit-step response for \(\tau=\frac{1}{3}\) and \(\frac{2}{3}\), plot the two curves versus \(0...
-
a. Identify the damping type and find the free response. b. Plot the free response by using the initial command. \(\ddot{x}+3 \dot{x}+4 x=0, \quad x(0)=\frac{2}{5}, \quad \dot{x}(0)=0\)

Study smarter with the SolutionInn App


