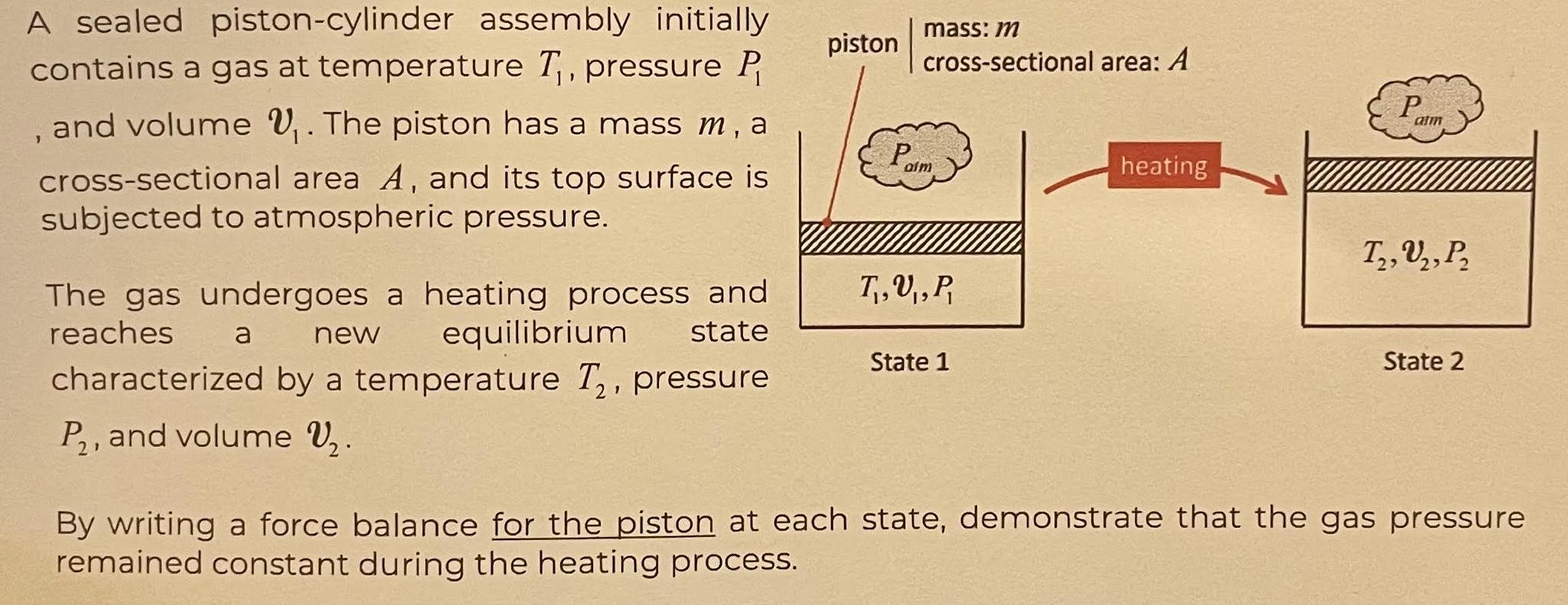
A sealed piston-cylinder assembly initially contains a gas at temperature T, pressure P , and volume...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A sealed piston-cylinder assembly initially contains a gas at temperature T₁, pressure P , and volume ₁. The piston has a mass m, a cross-sectional area A, and its top surface is subjected to atmospheric pressure. The gas undergoes a heating process and reaches equilibrium state a new characterized by a temperature T₂, pressure P₂, and volume V₂. piston mass: m cross-sectional area: A Paim T₁,U₁, P, State 1 heating P atm T₂, U₂, P₂ 2' State 2 By writing a force balance for the piston at each state, demonstrate that the gas pressure remained constant during the heating process. A sealed piston-cylinder assembly initially contains a gas at temperature T₁, pressure P , and volume ₁. The piston has a mass m, a cross-sectional area A, and its top surface is subjected to atmospheric pressure. The gas undergoes a heating process and reaches equilibrium state a new characterized by a temperature T₂, pressure P₂, and volume V₂. piston mass: m cross-sectional area: A Paim T₁,U₁, P, State 1 heating P atm T₂, U₂, P₂ 2' State 2 By writing a force balance for the piston at each state, demonstrate that the gas pressure remained constant during the heating process.
Expert Answer:
Answer rating: 100% (QA)
Lets start by writing the force balance for the piston in State 1 Ftop Fbottom Fatmos where Ftop is ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
A unit mass of an ideal gas at temperature T undergoes a reversible isothermal process from pressure P1 to pressure P2 while losing heat to the surroundings at temperature T in the amount of q. If...
-
Bruno Corp. has decided to expand its operations. The bookkeeper recently completed the following statement of financial position in order to obtain additional funds for expansion: Instructions (a)...
-
In a study of speed dating conducted at Columbia University, female subjects were asked to rate the attractiveness of their male dates, and a sample of the results is listed below (1 = not...
-
Prepare an ERD for a real estate firm that lists property for sale. Also prepare a definition for each entity type, attribute, and relationship on your diagram. In addition, draw a data model for...
-
The fugacity co-efficient of a substance is the ratio of its fugacity to the (a) Mole fraction (c) Pressure (b) Activity (d) Activity co-efficient.
-
Suppose that the contract had no liquidated damages provision (or the court refused to enforce it) and X Entertainment breached the contract. The breach caused the release of the film to be delayed...
-
Does Africa's colonial past affect its current economic position in the world? Why or why not? Back your points up with references only to Steger, Ellwood, Learning Materials, and resources below....
-
Call center A processed 1.2 million phone orders for a total of 2.4 million items. 50% of the orders are new customers and it takes 7 minutes to set up an order for a new customer. 50% of the orders...
-
During the COVID-19 pandemic, people saw major disruptions to supply lines throughout the country. Nowhere was this more evident than in the healthcare industry. Among the biggest impact was the...
-
Why are org charts commonly reviewed by internal auditors?
-
Nike Inc. (NKE) Nike Inc. reports 2012 net operating working capital of \$3,931 million and 2012 long-term operating assets of \(\$ 2,666\) million. a. Forecast Nike's 2013 net operating working...
-
If \(f(t)=e^{-3 t / 5}+t \ln (t+1)\), evaluate \(d f / d t\) when \(t=4.4\), by: a. Using the subs command. b. Conversion into a MATLAB function.
-
What are risk-neutral probabilities? How can they be used to value options?
-
Write a program that uses StdAudio and Picture to create an interesting two-dimensional color visualization of a sound file while it is playing. Be creative!
-
QUESTION 1 1. Boston Company has the following balances as of the year ended December 31, 2015. Direct Materials Inventory WIP Inventory Finished Goods Inventory Cost of Goods Sold Additional...
-
Periwinkle Company is a multinational organization. Its Parts Division is located in Lavender Land, while its Assembly Division is located in North Orchid. During the current year Periwinkle Companys...
-
The first line of the Balmer series occurs at a wavelength of 656.3 nm. What is the energy difference between the two energy levels involved in the emission that results in this spectral line?
-
How many molecules of ethane (C2H6) are present in 0.334 g of C2H6?
-
The phase diagram of sulfur is shown here. (a) How many triple points are there? (b) Monoclinic and rhombic are two allotropes of sulfur. Which is more stable under atmospheric conditions? (c)...
-
A system has the differential equations \(\left[\begin{array}{lll}5 & 0 & 0 \\ 0 & 3 & 0 \\ 0 & 0 & 2\end{array} ight]\left[\begin{array}{c}\ddot{x}_{1} \\ \ddot{x}_{2} \\ \ddot{x}_{3}\end{array}...
-
Lagranges equations are used to derive the differential equations for a three degree-of-freedom system resulting in \(\left[\begin{array}{lll}m_{11} & m_{12} & m_{13} \\ m_{21} & m_{22} & m_{23} \\...
-
The following data relate to the manufacturing activities of MilTech company during November: 1. Sales $400,000 2. Purchases $60,700 3. Expenses include: Light and power for manufacturing: $2,700 ...

Study smarter with the SolutionInn App


