A steel casting cools to 90% of the original temperature difference in 30min in still air....
Fantastic news! We've Found the answer you've been seeking!
Question:
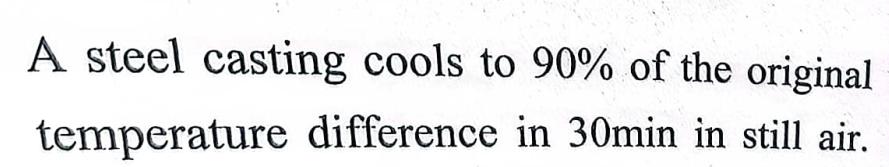

Transcribed Image Text:
A steel casting cools to 90% of the original temperature difference in 30min in still air. The same casting is cooled to 90% of the original temperature difference in a moving air stream. the convective heat transfer coefficient in the moving air is 5 times that of still air. the time in minutes to cool the casting in moving air is ................ The same casting is cooled to 90% of the original temperature difference in a moving air stream. The convective heat transfer coefficient in moving air is 5 times that of still air. The time (in minutes) to cool the casting in moving air is A steel casting cools to 90% of the original temperature difference in 30min in still air. The same casting is cooled to 90% of the original temperature difference in a moving air stream. the convective heat transfer coefficient in the moving air is 5 times that of still air. the time in minutes to cool the casting in moving air is ................ The same casting is cooled to 90% of the original temperature difference in a moving air stream. The convective heat transfer coefficient in moving air is 5 times that of still air. The time (in minutes) to cool the casting in moving air is
Expert Answer:
Related Book For 

Statistics The Exploration & Analysis Of Data
ISBN: 9780840058010
7th Edition
Authors: Roxy Peck, Jay L. Devore
Posted Date:
Students also viewed these chemical engineering questions
-
A counter flowing heat exchanger cools air at 600 K, 400 kPa to 320 K using a supply of water at 20C, 200 kPa. The water flow rate is 0.1 kg/s and the air flow rate is 1 kg/s. Assume this can...
-
A counterflowing heat exchanger cools air at 600 K, 400 kPa to 320 K using a supply of water at 20C, 200 kPa. The water flow rate is 0.1 kg/s and the air flow rate is 1 kg/s. Assume this can be done...
-
A steel casting weighing 2 kg has an initial temperature of 500oC; 40 kg of water initially at 25oC is contained in a perfectly insulated steel tank weighing 5 kg. The casting is immersed in the...
-
The death of Steve Jobs in 2011 sent the world into mourning for the loss of a great innovator. No longer would the world witness Apples enthusiastic CEO clad in his characteristic black turtleneck...
-
Accuzeit is a small New England company that manufactures custom clocks. It uses a job costing system that applies factory overhead on the basis of direct labor-hours. Budgeted factory overhead for...
-
Consider the following facts. During its assessment of the accounts payable department, the internal audit function identified the following observations: Inadequate segregation of duties over...
-
Cheung Keong Ltd. currently has \($2,150,000\) in current assets and \($860,000\) in current liabilities. The companys managers want to increase the firms inventory, which will be financed by a...
-
The balance sheet and income statement for Carver Enterprises, Inc. are as follows: Balance Sheet 2013 Cash and marketable securities .........$ 500 Accounts receivable .............. 6,000...
-
(a) An investor purchased 300 units of a Mutual Fund at Rs. 12.25 per unit on 31 December, 2009. As on 31st December, 2010 he has received Rs. 1.25 as dividend and Rs. 1.00 as capital gains...
-
Green Planet, Inc., a manufacturer of air filter systems for industrial facilities, is considering the addition of a new system to its current product line. The following data has been forecasted: ...
-
Which of the following is a deductible medical expense? A.)Funeral, burial, or cremation expenses . B.)Medicare Part B premiums withheld from the taxpayer\\\'s social security benefits. .)Payments...
-
Enter the following in the appropriate creditors' accounts (i.e. your suppliers' accounts) only. Do not write up the other accounts. Then balance-off each of these personal accounts at the end of the...
-
Redraft each of the accounts given in your answer to Review Question 5.1 in three-column style. 2016 May 1 Sales on time to B. Flyn 810; F. Lane 1,100; T. Fey 413. 4 Sales on time to F. Start 480; B....
-
On 1 March 2017, J. Blane Ltd, 7 Down Road, Middlefield, sold the following goods on credit to T. Roy & Son, Ballano Golf Club, Ringlee, Yorkshire: Order No. B/162 4 sets 'Silver Tiger' golf dubs at...
-
You are to enter up the sales, purchases, returns inwards and returns outwards day books from the following details, then to post the items to the relevant accounts in the sales and purchases...
-
A You are to enter the following items in the books, post to personal accounts, and show the transfers to the General Ledger. 2017 1 Credit purchases from: K. Hill 380; M. Norman 500; N. Senior 106....
-
Boulderado has come up with a new composite snowboard. Development will take Boulderado four years and cost $250,000 per year, with the first of the four equal investments payable today upon...
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
The stem-and-leaf display of Exercise 3.16 uses only four stems. Construct a stem-and-leaf display for these data using repeated stems 1H, 2L, 2H 4L. For example, the first observation, 29, would...
-
Researchers at the Hospital for Sick Children in Toronto compared babies born to mothers with diabetes to babies born to mothers without diabetes (Conditioning and Hyperanalgesia in Newborns Exposed...
-
Both r2 and se are used to assess the fit of a line. a. Is it possible that both r2 and se could be large for a bivariate data set? Explain. (A picture might be helpful.) b. Is it possible that a...
-
Allister Company uses both debt capital and equity capital to fund new projects. The before-tax cost of debt capital is 12 percent. The cost of equity capital is 10 percent. Allister's effective tax...
-
Use an Internet search engine to identify three companies or agencies that use present worth analysis to make business decisions as evidenced in their annual reports.
-
Use the Internet to access ConocoPhillips's most recent annual report. a. What evidence do you find that ConocoPhillips continues to rely on present worth analysis in its corporate decision making?...

Study smarter with the SolutionInn App



